the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Qualification of an online device for the measurement of the oxidative potential of atmospheric particulate matter
Albane Barbero
Guilhem Freche
Luc Piard
Lucile Richard
Takoua Mhadhbi
Anouk Marsal
Stephan Houdier
Julie Camman
Mathilde Brezins
Benjamin Golly
Jean-Luc Jaffrezo
Gaëlle Uzu
Particulate Matter (PM) and gaseous pollutants can carry or induce the production of Reactive Oxygen Species (ROS) in the lung environment, causing oxidative stress, a key factor in the development of cardiovascular and pulmonary outcomes. Over the past decade, numerous techniques have been implemented to assess the Oxidative Potential (OP) of aerosols, i.e., their ability to oxidise the lung environment as an initial proxy of subsequent biological processes. Offline measurements from filters collected from air samplers are widely assessed but are probably underestimating PM redox activity due to the short lifetime of several ROS and/or the loss of the most volatile compounds on filters in a non-proportional and unsystematic way. This study introduces a new device, called ROS-Online, allowing the automatic and near real time measurement of two complementary OP assays, OP Ascorbic Acid (OPAA) and OP Dithiothreitol (OPDTT), sensitive to ambient PMs at mass concentrations about [PM10] ∼ 20 µg m−3. The ROS-Online device is designed to reproduce the exposure and interaction of airborne particles with the respiratory system. ROS-Online consists of three main modules: (i) an air sampling module using a BioSampler® to collect airborne PM, (ii) a distribution module that transports samples and reagents to (iii) a measurement module that relies on spectrophotometric methods to monitor chemical reactions in real time. Its operation is based on established OPAA and OPDTT protocols, ensuring comparability with existing offline OP measurement methods. Compact and transportable (75 × 65 × 170 cm, 85 kg), ROS-Online is designed for deployment in air quality monitoring stations and allows for autonomous operation over 2 weeks. With a high particle collection efficiency (> 90 % by mass for PM1 and PM2.5) and greater sensitivity than offline methods, it provides accurate and reliable results across a wide range of aerosol concentrations, from urban backgrounds to highly polluted environments. The qualification of the device demonstrated an excellent correlation with offline methods for both OPAA and OPDTT measurements (r > 0.96), over positive controls, confirming the reliability and specificity of ROS-Online for continuous atmospheric aerosol OP monitoring. ROS-Online was deployed in the field, in an urban background site, where OPAA of ambient air was measured for 15 continuous days and OPDTT for 6 continuous days. Results showed a good correlation with ozone (O3) signal (R2 = 0.74), underlying the importance of considering pollutants' interaction in OP measurements, as laboratory experiment showed no OP response when introducing O3 alone into the instrument. Comparison of ROS-Online measurements with established offline methods showed an excellent correlation for both AA and DTT assays (r > 0.96), supporting its reliability for atmospheric monitoring. These preliminary results mark an important step towards establishing ROS-Online as a viable and effective tool for OP assessment in future research and monitoring endeavours.
- Article
(2856 KB) - Full-text XML
-
Supplement
(1664 KB) - BibTeX
- EndNote
Air pollution has emerged as a critical public health issue and its exposure is considered to be the second most important risk factor for the total death rate for all sexes and ages in 2021 (State of Global Air Report 2024). An estimation was made of 8.1 million premature deaths worldwide in 2021, attributed to poor outdoor and household air quality due in large part to the significant contribution of particulate matter (PM) (WHO, 2023). This finding is drawn from cross-sectional studies based on mass concentrations of particulate matter (PM) in ambient air combined with health and mortality data. A significant number of epidemiological studies have linked some cardio-respiratory diseases and cancers, such as heart disease, stroke, diabetes, lung cancer, and chronic obstructive pulmonary disease to particulate matter exposure (Manigrasso et al., 2020; Manisalidis et al., 2020; Qu et al., 2017; Turner et al., 2020). While the complete mechanisms are not yet fully understood, studies point out that PM is responsible for generating oxidants in vivo and inducing inflammation, thereby leading to cellular damage (Møller et al., 2014, 2020). More specifically, these mechanisms are driven by oxidative stress and involve reactive oxygen species (ROS) carried or induced by PM (Campbell et al., 2019; Delfino et al., 2013; Strak et al., 2012), and redox-active transition metals which, depending on their oxidation state, may participate in ROS production or act as direct oxidants, but also by certain trace gases (Dovrou et al., 2021). Trace metals are also recognized contributors to oxidative stress due to their catalytic redox properties. Such mechanisms and rates of ROS production rely on the composition of the aerosol such as the presence of oxidised polyaromatics hydrocarbons (OPAH), organic peroxides (ROOR) or transition metals. Recent advances in the field have emphasized the role of short-lived reactive species in OP (Campbell et al., 2025) and the impact of aerosol acidity and ligand-mediated metal solubility on OP (Shahpoury et al., 2019, 2021, 2024a, b). Furthermore, newer antioxidant assays are being explored to complement traditional metrics (Shahpoury et al., 2019). Air quality monitoring policies are currently based on the measurement of PM total mass, but several studies invite to consider other parameters in future regulations since other physico-chemical parameters, among which, chemistry, size distribution, surface area, etc., broadly influence PM toxicity (Park et al., 2018; Song et al., 2021; Wittmaack, 2007). During the last decade, atmospheric scientists have been developing Oxidative Potential (OP) as a metric accounting for the redox and catalytic properties of PM (Calas et al., 2017; Cho et al., 2005). Recent studies demonstrated that at least in Europe, the OP of PM is driven by anthropogenic sources whereas the PM mass is mainly controlled by secondary inorganic components and crustal material, highlighting a different vision of the sources' impacts when using PM mass or PM OP (Daellenbach et al., 2020; Weber et al., 2021). Daellenbach et al. (2020) and Leni et al. (2020) also showed that the associations between pro-inflammatory lung biomarkers and PM2.5 were higher for PM samples with the highest OP levels. Those findings strongly promote the health relevance of the OP metric. The new directive 2024/2881/CE was adopted on 20 November 2024 for the modification of the Directive 2008/50/EC on air quality in Europe, and specifically addresses the measurement of OP in airborne particulate matter. This new directive emphasizes on and recommends the measurement of OP as part of the broader assessment of air quality impacts on human health.
In the last 20 years, acellular methods have been developed to assess the redox activity of PM through various probes (Bates et al., 2015; Calas et al., 2018; Rao et al., 2020). Most of them rely on the depletion's quantification of a lung antioxidant, or surrogate, when in contact with PM. These traditional offline methods are widely used, and although they have a good recovery efficiency of the particulate matter, they involve bias in the estimation of OP. First, these methods severely underestimate PM redox activity due to the very short lifetime of some ROS (such as hydroxyl radical •OH) and/or the loss of the most volatile compounds (such as formaldehyde HCHO) in a non-proportional and unsystematic way (Campbell et al., 2025; Jiang et al., 2019). Second, they do not allow near-real-time OP estimation, nor the OP analysis of soluble gases (Carlino et al., 2023). To overcome such drawbacks of offline methods, semi-continuous prototypes for the OP measurement of solubilised PM filter samples and automatic on-site devices have recently been developed.
Indeed, semi-automatic laboratory methods for ROS analysis were developed based on the well-known dithiothreitol (DTT), ascorbic acid (AA) and dichlorofluorescin (DCFH) tests (Fang et al., 2015; King and Weber, 2013; Yu et al., 2020). Fuller et al. (2014) have designed a fully automated, portable and integrated DCFH technique, allowing the PM sampling and solubilisation before their subsequent analysis (Fuller et al., 2014). Ambient air measurements at a London urban site confirmed that this device was sensitive enough to measure ambient ROS in a European urban environment, and showed the relevance of such instrument into air quality measurement stations (Wragg et al., 2016). More recently, two separate prototypes were developed to assess online OP using fluorescence methods, one based on the OPAA assay and the other on the OPDCFH assay (Campbell et al., 2019; Utinger et al., 2023). The sensitivity of this device was tested on several transition metals, biogenic and anthropic secondary organic aerosol, and a mix of them, concluding that the method allowed for OP measurement in polluted urban environments (Campbell et al., 2023). Puthussery et al. (2018), showed a quasi-continuous data set over 2.5 months of OPDTT and in a recent paper by Campbell et al. (2024) a more or less continuous OPAA data set over three months is shown. Nevertheless, the different OP assays being sensitive at varying levels to diverse ROS-generating compounds (Lin et al., 2022), using several assays is crucial to provide a wider picture of the redox processes at stake.
The ROS-Online device, a stand-alone prototype for near-real-time measurement of ambient air OP via two complementary assays, is currently under development at the Institute for Environmental Geosciences (IGE, Grenoble, France). The device integrates two independent measurement lines, allowing the simultaneous measurements of ascorbic acid OP (OPAA) and dithiothreitol OP (OPDTT). Both tests are known for their physiological, practical, and economic advantages, and are widely used in offline methods (Dominutti et al., 2025; Mudway et al., 2004; Perrone et al., 2019; Rao et al., 2020). This device differs from others by assessing both OPAA and OPDTT from a unique atmospheric sample collected through a BioSampler® (SKC) for both soluble gases and particulate matter.
The present paper first presents in Sect. 2 the ROS-Online device and its main characteristics and operating principles. Section 3 is devoted to the sampler characterisation and ROS-Online's response evaluation in a simulated polluted environment with controlled airflow conditions. Section 4 presents the calibration of the two OP assays of ROS-Online, including an intercomparison with offline measurements, the latter being routinely implemented at IGE (e.g., Calas et al., 2017, 2018; Dominutti et al., 2023, 2025; Weber et al., 2018). Section 5 presents atmospheric measurements using ROS-Online under real conditions at a traffic site.
2.1 Description and main characteristics
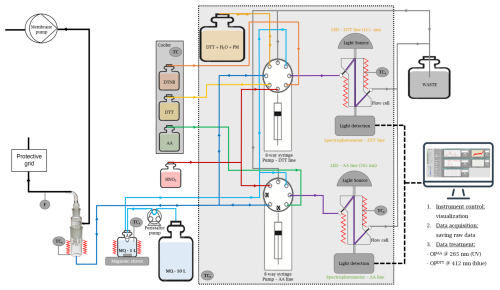
ROS-Online simulates pulmonary exposure and interaction with ambient air, aiming to replicate the physiological processes of inhalation and respiratory surface contact. The device is patented (PCT PCT/EP2021/080824, number WO2022096675, 12.06.2022. Priority: FR 20 11431 ROS ONLINE). The operating and measurements principles rely on the existing offline protocols for OPAA and OPDTT. This choice will allow to compare the overall body of data already available for offline OP measurements with that produced by ROS-Online. The prototype is described in Fig. 1 and consists of three main modules: (i) the sampling module for the ambient air collection, (ii) the distribution module, and, (iii) the measurement module for chemical reaction monitoring. An additional computer is used for managing the device and all data processing steps using LabVIEW application.
2.1.1 Offline method and adaptation to ROS-Online
The offline method originally adapted from Cho et al. (2005) and Li et al. (2009a) is fully described in Calas et al. (2017) and Weber et al. (2018, 2021). Briefly, particulate matter (PM) extracts from atmospheric filters (150 mm-diameter pure quartz fibre filters) during 75 min are introduced into two 96-well plates maintained under physiological conditions (pH = 7.4, T = 37 °C). To ensure homogeneity, the mixtures are shaken for 60 s for DTT and 10 s for AA. The absorbance of the matrix is read after 3 s for DTT and 10 s for AA. The reactions are initiated by adding AA or DTT to the respective plates, allowing the antioxidants to react with PM-induced reactive oxygen species (ROS). AA depletion is monitored continuously over 30 min at λ = 265 nm using a plate reader spectrophotometer (TECAN, model M1000 Infinite), and the depletion rate is determined by linear regression. DTT depletion is assessed indirectly by titrating the remaining DTT with dithionitrobenzoic acid (DTNB) at specific time points (0, 15, and 30 min). This reaction produces 5-thio-2-nitrobenzoic acid (TNB), which absorbs at λ = 412 nm and is measured using another plate reader (TECAN, model M200 Infinite). Both AA and DTT depletion are quantified using the Beer–Lambert law (Eq. 1):
where A is the absorbance; ε is the molecular extinction coefficient [L mol−1 cm−1]; L is the optical path, i.e., the wells' depth for the off-line method; c is the followed antioxidant concentration (AA) or the concentration of its reaction product (TNB) in the flow cell [mol L−1], I is the transmitted intensity of the solution; I0 is the intensity at t = 0 min. Both AA and DTT consumption rates, ω, are calculated following Eq. (2):
where s is the slope value of the linear regression of the measured absorbance over time [s−1] ; A0 is the absorbance at t = 0 min; and η0 is the initial quantity of reagent [mol]. Blanks are performed using filter and field blanks to account for absorbance due to quartz fibre fragments or potential contamination.
The ROS-Online system automates the previously described offline assays for continuous ambient air monitoring. The flow-based system replicates the essential steps of the offline protocol while enabling time-resolved measurement of oxidative potential (OP).
In the AA line, the AA solution and PM sample are first mixed during withdrawal into the syringe pump. This mixture is then transferred into the flow cell (FC), where the AA depletion is monitored by UV absorbance at 265 nm for 10 min. In the DTT line, since DTT does not absorb light directly but its reaction product TNB does, the DTT and sample are mixed in the syringe pump and stored in a reaction reservoir for 15 min. After this incubation, DTNB is added (again via mixing in the syringe pump), producing TNB. This final mixture is introduced into the FC, where absorbance at 412 nm is recorded to quantify the remaining DTT indirectly. For both lines, the Beer-Lambert law (Eq. 1) and the consumption rate formula (Eq. 2) are applied, with the optical path L corresponding to the flow cell length in this case. Ultra-pure water (Type 1 Milli-Q®) is used in place of PM extract for blanks.
The oxidative potential of ambient air is calculated using Eq. (3), following the approach by Fang et al. (2015, 2016):
where Vs is the ambient air volume sampled by ROS-Online; VFC the FC volume; and VBS the final sampled volume, considering evaporation in the sampling device, i.e. a modified BioSampler® (U.S Patent No. 5,902,385). The BioSampler® was specifically modified by a glassblower to include a custom-fabricated bottom outlet, facilitating automated extraction of the collected liquid sample. VBS is calculated from the initial and final volumes measured via humidity and temperature sensors. Dilution due to reagent addition is negligible but accounted for via volume correction in data processing.
The sensitivity of ROS-Online enables one complete measurement of both OPAA and OPDTT over a 20 min sampling period, repeated every 60 min in moderately polluted environments such as European urban background sites. ROS-Online is designed as a mobile, rack-mounted instrument (75 × 65 × 170 cm; 85 kg), requiring ∼ 800 W of power and an operating environment of 15–30 °C. All components are temperature-controlled. The system is field-deployable and can be operational within a few hours. The current design supports autonomous operation for approximately 2 weeks, limited by ultra-pure water consumption and storage.
2.1.2 Prototype features
Airborne PM and soluble gases are collected into a BioSampler® filled with 20 mL MQ water by pumping ambient air at a constant flow rate of ∼ 10.5 L min−1. This airflow is monitored by using a Venturi flow meter. The BioSampler® nozzle section contains three tangential 0.630 mm nozzles that act as sonic orifices that maintains a pressure drop of ∼ 0.5 atm or more across the sampler at normal atmospheric conditions (sonic flow) (BioSampler, 2024). Nebulisation of the water is generated via the nozzles during vacuuming ensuring optimal gas/liquid exchange and homogenisation of the solution. No physical filters are used inside the BioSampler®; particles are directly collected into MQ water. The BioSampler® is kept in a controlled chamber at a temperature of 37 °C. During sampling, a fraction of the water in the BioSampler® reservoir can evaporate due to the temperature differential between the ambient air and the thermostated sampling chamber (37 °C). To monitor this, ROS-Online is equipped with temperature and relative humidity sensors at the inlet and outlet of the BioSampler®. These data allow the estimation of the actual volume of liquid remaining at the end of the sampling period. Since the oxidative potential (OP) is expressed per cubic meter of air sampled (Eq. 3), the corrected final sample volume is essential for accurate post-processing and normalization. It is important to note that DTT as well as AA and DTNB are added after the sampling step, during the controlled reaction phase, and always in fixed volumes. Therefore, evaporation during sampling does not affect the reagents concentrations in the reaction, but only the total sampled air volume used to calculate OP. The device requires fluids for air sampling, rinsing between each measurement, and to perform OPAA and OPDTT measurements following protocols described above. A 20 L Jerry can (GDPE Nalgene®) of MQ water automatically feeds a heated glass tank (1 L) that delivers water for collection and reactions at physiological temperature, this tank is kept on a magnetic stirrer to ensures homogeneous temperature distribution and prevents stratification. Tanks containing AA (185 µM), DTT (2.12 mM) and DTNB (2.12 mM) solutions, being in phosphate buffer at pH = 7.4, are sheltered from the light and stored at T = 4 °C in a cooler and refreshed every 10 to 12 d. The rinsing solution (HNO3 0.1 % in MQ water) is stored in 2 L tank. All fluids are distributed by two 8-channel syringe pumps (PUMP Cadent 6 48K Level 3 – Norgren) via PTFE tubing (1/16′′). In the AA line, the AA solution and the sample are first mixed inside the syringe pump during withdrawal. The resulting mixture is then introduced into the flow cell (FC), where the reaction is monitored by spectroscopy at 265 nm over 10 min. In the DTT line, since DTT itself is not absorbing but its reaction product (TNB) is, the DTT and sample are also first mixed in the syringe pump and then transferred to a reservoir to allow the reaction to proceed for 15 min. Subsequently, DTNB is added – again mixed with the reactive mixture in the syringe pump – and the resulting TNB is measured in the FC for absorbance measurement at 412 nm.
2.1.3 Spectrophotometric monitoring of the reaction
Each line of measurement has its own {light source + spectrophotometer} couple: {ILR-ZZ01-Z265-LS0xx-SC201 (Stanley) + MAYA 2000 Pro (Ocean Optics)} and { M415F3 (Thorlabs) + MAYA 2000 Pro (Ocean Optics)} for the AA and DTT assays respectively. For kinetic monitoring of the reaction, AA + PM and DTT + DTNB + PM mixtures are pushed respectively into two microfluidic Z-flow cells (100 mm PEEK − IDIL), equipped with pressure controllers to avoid bubble formation that could compromise the absorbance measurement.
2.1.4 Comparison with other online devices
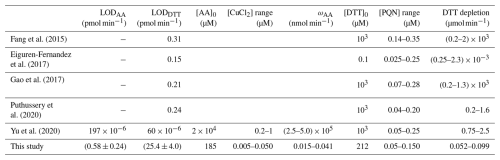
This section is intended to highlight how ROS-Online compares with existing instruments previously deployed for real-time OP measurements. Several automated and semi-automated prototypes characterising OP have been reported in the literature. Fang et al. (2015) and Gao et al. (2017) adapted the widely used OPDTT measurement, while Campbell et al. (2019) proposed a fully automated prototype based on AA chemistry following the design presented in Wragg et al. (2016). Eiguren-Fernandez et al. (2017) developed an online monitor which combined a liquid spot sampler and a chemical module optimized for online OPDTT measurements. Finally, Yu et al. (2020) set a multi-endpoint analyser including both OPAA and OPDTT assays. It should be noted that initial concentrations of reagents differ from one study to another as well as some of the experimental conditions. For example, the solubilisation medium used to solubilise airborne PM do not present the same chemical composition for each device (see discussion below). As shown in Table 1, comparison for both assays with other developed prototypes concludes to a much better background noise or blank for ROS-Online. The limit of detection (LOD) of ROS-Online was defined as LOD = ωblank−mean + 3 σ, where σ is the standard deviation calculated over n measurements of pure MQ water blanks (Fang et al., 2015; King and Weber, 2013). ROS-Online LODs for OPAA and OPDTT assays in standard operation conditions were (0.58 ± 0.24) pmol min−1 (n = 342 over a period 15 d) and (25.4 ± 4.0) pmol min−1 (n = 118 over a period of 6 d), respectively. In comparison, LOD for IGE's offline methods were (6.70 ± 0.78) pmol min−1 (OPAA assay, n = 36) and (19.93 ± 3.01) pmol min−1 (OPDTT assay, n = 36). ROS-Online is about twice more sensitive in measuring OPAA than the offline IGE method and exhibits an equivalent sensitivity than the OPDTT assays in the already published online methods. As shown later, such a sensitivity allows OP measurements over 20 minutes in operating conditions for the European environments we studied. Calibration experiments with artificial solutions of PM redox components like CuCl2 or phenantroquinone (PQN) (more information in Sect. 4) show that ROS-Online exhibits linearity over a high dynamic range of concentrations for these species. This is possible because ROS-Online allows large antioxidants depletions, i.e., high value of s in Eq. (2), from few pmol min−1 to thousands of pmol min−1 or hundreds of pmol min−1 for AA and DTT assays, respectively. This result let us assume that ROS-Online could be employed in very polluted environment (industrial sites, mining industry, smog cities etc.). The comparison with consumption rates (ω) achieved for calibrations in other studies (Table 1) highlights the influence of reactant concentrations on OP values. The signal to noise ratio (SNR) of ROS-Online is calculated for each assay following Eq. (4):
where It is the intensity (arbitrary units) of the spectrophotometric measurement of the sample at different times: t = 0 – reference, only MQ water is introduced in the measuring cells; t = 1 – start, the sample is introduced in the FC and the intensity is monitored after 60 s of reaction; and t = 2 – end, the intensity read at the end of the reaction, i.e., at t = 10 min for AA and t = 15 min for DTT. σI is the standard deviation on the intensities over 6 d of atmospheric monitoring. The maximum SNRs measured for AA and DTT assays were (39 ± 2) and (23 ± 5), respectively. Therefore, ROS-Online presents good sensitivity and low limit of detection (LOD), allowing to accurately quantify small variations in OPAA and OPDTT responses in near-real-time measurements of atmospheric PM.
A laboratory test-bench was used to characterize the response of the ROS-Online to atmospheric components under semi-controlled operating conditions. This was performed on a bench from LOCIE (Building Energy Processes Laboratory) in University of Savoie-Mont Blanc, Chambery, France. These tests aimed at evaluating the ability of ROS-Online to capture PMs, by studying the collection efficiency of the BioSampler® and will be compared to results obtained with the same device in recent studies (Bøifot et al., 2024; Lin et al., 2018; Mescioglu et al., 2021). Measurements with specific particles directed to the inlet of the instrument were achieved to evaluate the capability of ROS-Online to detect and measure ambient OP in near real atmospheric conditions. Figure 2 shows a schematic representation of the experimental set-up made of stainless-steel pipes (internal diameter 72 mm). The carrier gas used is ambient air from the laboratory, pre-filtered by a HEPA (U15 -EN1822- with a MPPS, or most penetrating particle size = 99.9995 %) filter and the air flow (FI) is controlled by a variable speed fan allowing a flow rate from 5 to 70 m3 h−1. The bench is also equipped with thermo-hygrometers measuring the temperature (TI) and relative humidity (RH) during experiments. Finally, the potential impact of oxidising gases on the ROS-Online response was also studied by injecting variable O3 concentrations (in the 0–50 ppb range) directly at the sampling inlet of the ROS-Online system.
3.1 BioSampler® characterization of collection efficiency
A first characterization of PM collection efficiency is carried out with the BioSampler® only, by sequential opening of the three-way valve (Fig. 2 – ways A and B) during sampling times of 15 min. Particle size distribution and PM concentration within the bench are monitored using two devices: an optical PM counter (particles range between 0.3 and 20 µm – model 1.108, Grimm) and a portable scanning mobility particle sizer, NanoScan® SMPS (nanometric particles 10 to 300 nm – model 3910, TSI Inc.) equipped with a X-ray pre-neutralization step (model 3088, TSI Inc.) prior to the sample analyses (Fig. 2). An aerosol of liquid particles is generated using a nebulizing particle generator (model ATM220, Topas) supplied with dry and filtered air. A 1 g L−1 KCl solution was used to generate a saline aerosol with normalized size distribution of particles. Relative humidity and temperatures were 40 % and 25 °C, respectively, the atomisation rate was set at 1 mL min−1 (or 6.0 × 10−5 m3 h−1), and the bench air-flow was 20 m3 h−1.
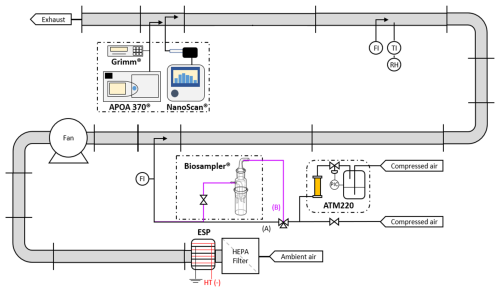
Figure 2Schematic diagram of the experimental bench at LOCIE laboratory: the figure shows the set-up for the BioSampler® collection efficiency experiment.
This methodology has been commonly used at LOCIE laboratory and described in detail in Chen et al. (2020). Shortly, the measurement of PM concentration for each ith class of diameter (n = 3 replicates) was performed between 20 nm and 5 µm. Firstly, a bypass of the BioSampler® (way-A) was used to determine the initial injected PM concentration which was subsequently measured after the air flow passed through the BioSampler® following the B-way (pink). The difference of particle number measured by these two ways allowed to calculate the single-pass fractional collection efficiency η(dp) for each class of diameter, which is defined by:
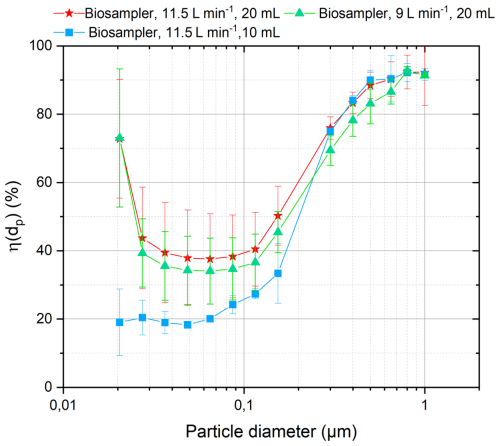
where NA,i and NB,i are the number of particles (cm−3) measured for each class of diameter through A- and B-ways, respectively. The fractional collection efficiency at different flow rates calculated using Eq. (5) are reported in Fig. 3. It shows U-shaped curves which are typically reported in literature for this type of bio aerosol sampler, thus our results are consistent with previously reported BioSampler® collection efficiencies (Bøifot et al., 2024; Guo et al., 2024; Su et al., 2020).
In this study, efficiencies of BioSampler® were tested at sampling flow rates of 9 and 11.5 L min−1 in order to frame the ROS-Online operating value of 10 L min−1. At 11.5 L min−1, the BioSampler® was filled with 10 or 20 mL of MQ water to evaluate the influence of evaporation. Similar results were obtained for both flow rates, with fractional collection efficiencies η(dp) in the range (34 ± 9.7) %–(37.6 ± 14.6) % at 65 nm and in the range (88.5 ± 3.5) %–(100 ± 7.0) % above 500 nm. The low fractional collection efficiencies (η(dp) ∼ 40 %) for particles in the range (50–80) nm are similar to the results reported by Li et al. (2018). In the size range of 0.5 to 5 µm, the η(dp) above 90 % are also consistent with the literature values (Bøifot et al., 2024; Guo et al., 2024; Su and Vincent, 2004). PM2.5 collection efficiencies by mass, recalculated from number distributions, were close to 82 % to 85 %. Indeed, KCl atomization produces relatively few large particles, with approximately 70 % of the total mass carried by particles larger than 300 nm. In contrast, ambient atmospheric aerosols are typically dominated by accumulation and coarse mode particles, with studies showing that more than 90 % of the mass is often found in particles larger than 300 nm in urban background or remote environments (Brock et al., 2021; Keuken et al., 2013). Therefore, the reduced collection efficiency observed for particles smaller than 0.3 µm is expected to result in only a limited loss of total collected mass across the 10 nm to 10 µm range. However, we acknowledge that in specific environments such as traffic sites, where freshly emitted ultrafine particles can contribute more significantly to PM mass, this limitation may be more relevant (Cheng and Lin, 2010; Gillies et al., 2001).
As shown in Fig. 3 for experiments with different sampling volumes, the loss of sampling liquid through evaporation also reduces the collection efficiency, especially for nanometric particles (< 100 nm). This is likely because of a lower aerosolisation of the collection liquid when the distance between the inlet nozzles and the liquid surface increases. These results suggest that the collection efficiency for nanometric particles may significantly decline over prolonged sampling durations, thereby supporting the choice to fill the BioSampler with 20 mL of ultrapure (MQ) water in subsequent measurements.
3.2 Atmospheric OP with semi controlled PM generation
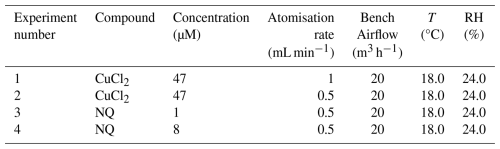
Four experiments were conducted with artificial solutions of CuCl2 and Naphthoquinone (NQ), both being major redox components of PM (Ayres et al., 2008; Charrier and Anastasio, 2012; Kumagai et al., 2002) to evaluate the ROS-Online response and to highlight the difference in reactivity of both probes. Experimental conditions are summarized in Table 2. ROS-Online collected over 20 min at 0.6 m3 h−1 (∼ 10.5 L min−1) inside the main air flow at 20 m3 h−1. An isokinetic calculation was performed to adjust the same velocity in the gas stream and in the ROS-Online sampling line. Laminar flow conditions were ensured by the distances between the disturbance zones and the tapping points, i.e., 3 times the pipe diameter upstream and 10 times the pipe diameter downstream. The size distribution of the particles generated during the four experiments is presented in Fig. S1 of the Supplement. Experiments 1 and 2 were conducted with a 47 nM CuCl2 solution atomised in the experimental bench at two different rates, 1 and 0.5 mL min−1, respectively, in order to achieve different concentration levels of PMs. Indeed, between experiments 1 and 2, PM concentrations changes from (64 ± 10) to (34 ± 5) µg m−3 for PM2.5 and from (59 ± 9) to (30 ± 5) µg m−3 for PM1. Experiments 3 and 4 were conducted using 1 and 8 mM solutions of NQ respectively, leading to PMs concentration levels of (215 ± 18) to (364 ± 18) µg m−3 for PM2.5 and (179 ± 6) to (323 ± 13) µg m−3 for PM1. Regardless of the concentration of the solutions or the atomisation rate, the nebulized particle generator produces a majority of PM1, with an average of (88 ± 4) % of the particles by mass having a diameter of 1 µm or less.
Figure 4 shows the results for the different experiments on both assays of ROS-Online.
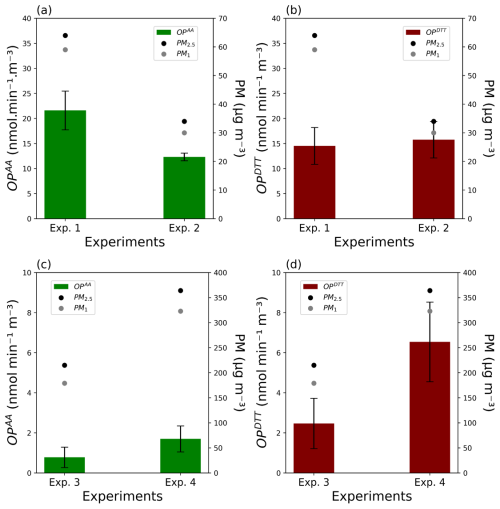
Figure 4(a, c) OPAA and (b, d) OPDTT results obtained during the four experiments carried out in the experimental bench. Dots represent the average PM2.5 (black) and PM1 (grey) mass concentrations (µg m−3) in the main flow, bars represent the OP ± 1σ (nmol min−1 m−3) of AA (green) and DTT assays (brown).
As shown in Fig. 4a and b for CuCl2 experiments 1 and 2, OPAA confirmed its high metal-sensitivity (Calas et al., 2018; Fang et al., 2016; Godri et al., 2011) with OP values of (21.6 ± 3.9) and (12.3 ± 0.8) nmol min−1 m−3. This result is consistent with the commonly reported sensitivity of ascorbic acid (AA) to transition metals. Although the oxidation of AA itself does not proceed via Fenton-type radical reactions, the superoxide radicals generated during its oxidation can subsequently react with iron through Fenton chemistry (Bates et al., 2019; Fang et al., 2016; Bresgen and Eckl, 2015; Pietrogrande et al., 2022). OPDTT results show lower variability across experiments, (14.5 ± 3.7) and (15.8 ± 3.6) nmol min−1 m−3, respectively, suggesting a less concentration-dependent response than AA, as already evidenced in the literature (Charrier and Anastasio, 2012; Lin and Yu, 2011; Pietrogrande et al., 2022a).
PM2.5 and PM1 concentrations in the bench are divided by a factor of two between experiments 1 and 2. Interestingly, rather different ratios of 1.8 and 0.9 were observed for OPAA and OPDTT between the two experiments. Thus, the response of the DTT assay to copper particles seems to reach a maximum, regardless of their mass concentration. However, it remains unclear whether this represents an absolute maximum for OPDTT or a local plateau that could rise further if concentrations still increases. This observation could be explained by the intricate two-stage kinetics of the reaction between DTT and oxygen in the presence of copper which, as already observed for lead (Uzu et al., 2011), involves the formation of a [DTT-Cu] complex (Kachur et al., 1997) and thus limits the redox reactivity between the two compounds.
Regarding NQ experiments 3 and 4 (Fig. 4c and d), PM2.5 and PM1 concentrations in the bench are not proportional to the concentration of the solutions used for nebulization. Indeed, while an 8 times more concentrated solution is used for experiment 4, the ratio of PMs concentrations between the two experiments does not exceed 1.7 and 1.8 for PM2.5 and PM1, respectively. Considering OP values, OPAA appears to be less sensitive to NQ than OPDTT in these conditions, with OPAA values of (0.8 ± 0.5) (Exp. 3) and (1.7 ± 0.7) (Exp. 4), and OPDTT values of (2.5 ± 1.3) (Exp. 3) and (6.5 ± 2.0) nmol min−1 m−3 (Exp. 4). This has already been observed by Pietrogrande et al. (2022). Indeed, DTT is reactive to both organic and inorganic compounds and its high reactivity towards quinones has been highlighted many times (Calas et al., 2018; Jiang et al., 2020; Yang et al., 2014). However, both assays show an increase in OP values with PM concentrations, even though this increase is not proportional with ratios of 2.2 and 2.6 for OPAA and OPDTT, respectively. A non-proportional increase in PMs concentrations could be explained by many parameters such as the non-controlled NQ aerosol generation with the nebulized particle generator as organic aerosols like naphthoquinones might interact with stainless steel surfaces under varying conditions (Sherif and Park, 2006; Walker et al., 2022).
3.3 ROS-Online interference with ozone
Dovrou et al. (2021) highlighted the key role of ozone in the formation of ROS like H2O2, •OH, •O, or H•O2. Additionally, Stevanovic (2017) have demonstrated the presence of ROS in both gaseous and particulate phases of vehicular emissions. To test for this hypothesis, ROS-Online sensitivity to an oxidant gas such as O3 was explored in controlled conditions. In these experiments, O3 delivered by a 2B Tech O3 generator was introduced into the air stream sampled by ROS-Online, and a Thermo Scientific Model 49i O3 analyser was used to measure O3 levels at the exhaust of ROS-Online. This set-up allowed 100 % of the O3 introduced into the ROS-Online's airflow to be collected during sampling. O3 was introduced at concentrations ranging from 0 to 50 ppbv resulting in quite uniform AA depletions (Fig. S2) with mean and median values of (4.2 ± 1.1) and 4.0 pmol min−1, respectively. Therefore, ROS-Online's response seems not be correlated influenced by the ozone concentration present in the atmosphere. Indeed, the oxidative potential of O3 is not effectively captured via AA depletion in ROS-Online, potentially because: (1) ozone adsorbs or reacts with surfaces (e.g. pipes) before entering the sampler, or (2) ozone is not very soluble in water and/or the contact time between water and air is insufficient, so it cannot be trapped. In atmospheric conditions, it tends to adsorb/react with surfaces or airborne particles before it can dissolve in a liquid-phase assay (Bates et al., 2019; Bellini and De Tullio, 2019; Chang et al., 2021; Charrier and Anastasio, 2012). Other atmospheric oxidants, including more water-soluble inorganic and organic gaseous compounds, could also contribute to AA or DTT depletion, and further investigations are required to evaluate their role in the overall oxidative potential of the gas phase, as well as to assess the possible cross-sensitivity to ozone.
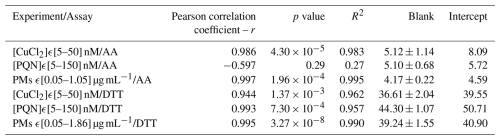
CuCl2 ([5–50] nM) and PQN ([5–150] nM) standard solutions were selected to assess the OPAA and OPDTT sensitivities, respectively, of the system. Concentrations ranges for Cu(II) and PQN were chosen to be representative of concentration levels measured in the atmosphere of European cities, i.e., CuCl2 [0.013–0.31] µg m−3 and PQN [0.0013–0.1619] µg m−3 (Delgado-Saborit et al., 2013; Denier van der Gon et al., 2013; Liu et al., 2024; Yang et al., 2018). Extract solutions from two real PM samples, collected in the peri-urban area of Grenoble and at a traffic site in Bern (Switzerland) were also analysed, for OPDTT and OPAA, respectively. Both samples were collected on pre-baked quartz filters (Pall, Tissuquartz) with a high-volume sampler (Digitel, DA80, 30 m3 h−1) equipped with a PM10 inlet. The filters were extracted in MQ water in a multi-tube vortex at 37 °C. In order to avoid any clogging of the prototype lines by remaining fragments from the quartz filters, syringe filters (Sartorius, 0.2 µm) were used to filter the extracted samples. Calibration solutions in the [0.05–1.86] µg PM mL−1 range (equivalent to [5–180] µg m−3 atmospheric PM concentrations; Borlaza et al., 2022) for the Grenoble filter and in the [0.05–1.05] µg PM mL−1 range ([5–100] µg m−3 equiv. PM) for the Bern sample were prepared. Each standard solution was analysed for OP in triplicate and the three replicates were used to calculate the standard deviations for the online method. It is important to note that the limits of detection (LODs) presented in Fig. 5 correspond to the minimum detectable concentrations based on spiked samples, while the value of 20 µg m−3 mentioned in the abstract refers to the minimum ambient PM mass concentration required for reliable detection under real atmospheric sampling conditions. Blanks containing only MQ water were analysed prior the standard solutions and their values are reported in Table 3. Figure 5a–d show ROS-Online response (corrected for the blanks) over the CuCl2 and PQN ranges for both AA and DTT assays. Except for PQN in the AA assay (Fig. 5c), the calibration responses were found to be acceptably linear for the intended application, as supported by the R2 values, Pearson correlation coefficients, and p values reported in Table 3. While some deviations from perfect linearity are observed, the statistical indicators confirm that the linear fits are adequate for use with environmental and experimental samples.
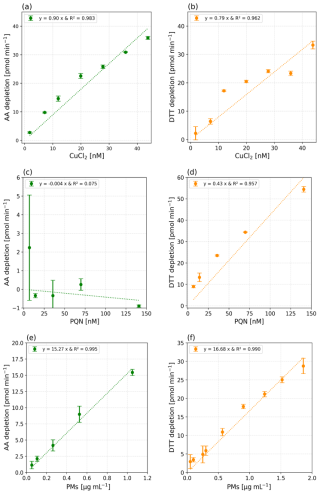
Figure 5Green dots represent the ascorbic acid consumption rate, i.e., depletion, in [pmol min−1] as a function of CuCl2 (a) and PQN (c) concentrations in [nM]. The orange dots represent the dithiothreitol depletion in [pmol min−1] as a function of CuCl2 (b) and PQN (d) concentration in [nM]. (e) Green dots represent the ascorbic acid depletion, in [pmol min−1] as a function of PMs samples [µg mL−1] taken from a traffic site filter (Bern), and (f) orange dots represent the dithiothreitol depletion in [pmol min−1] as a function of PMs samples [µg mL−1] taken from an urban background winter filter (Grenoble). Dashed lines represent the modelled linear regressions. Each sample was analysed in triplicate, and the figures present average (dots), with the error bars representing ± 1σ.
As shown in Fig. 5c, the AA response towards PQN over the [5–150] nM range seems null. This result is in accordance with Pietrogrande et al. (2022b), which demonstrated the sensitivity of OPAA to PQN for concentrations above 1 µM (at [AA]0 = 100 µM). It is therefore likely that [PQN] used here were too low for a response to be detected by ROS-Online. The relatively poor linearity observed in Fig. 5b is probably due to the low sensitivity of DTT to Cu(II). This result agrees well with what we observed during Exp. 1 and 2 at LOCIE bench (Fig. 4b). However, Fig. 5e and f show that the ROS-Online response is linear over the wide range of concentrations of PM extracts considered in this work i.e. from 0.05 to 1.86 µg PM mL−1 corresponding to PM ambient concentrations between 5 and 180 µg m−3. The response is highly linear on both assays, with determination coefficient R2 = 0.995 for AA (Bern sample) and R2 = 0.990 for DTT (Grenoble sample).
Table 3 reports the main characteristics of the linear regressions obtained with Cu(II) and PQN standard solutions and filter samples. Results are given without taking account the blank correction. As the intercept values are, to some extent, similar to the blank values, one can hypothesize that the non-zero intercept is likely related to the self-degradation of the antioxidants. This is supported by the fact that AA is sensitive to temperature and UV (Basak et al., 2023; Essodolom et al., 2020). In our protocol, the reacting solution containing AA is illuminated at 265 nm for 10 min in the FC and heated to 37 °C, thus explaining the self-consumption of AA in the ROS-Online device. Similarly, both DTT and TNB are also sensitive to light (Damodaran, 1985; Eyer et al., 2003). In that case, the solution in the FC is illuminated at 412 nm for 15 min, resulting in the self-degradation of these reactants and finally to non-zero intercepts we observed for the DTT assay. The consideration of this issue subsequently led us to systematically perform a blank for each real atmospheric measurement. The difference in blank values observed for AA (4 to 5 pmol min−1) and for DTT (37 to 44 pmol min−1) assays may be due to the quality of the solutions used, particularly of the ultrapure water used either for the standards solutions or for the PM extracts. Additionally, the difference in blank values observed between both assays is probably due to the kinetics itself (reaction time, wavelength etc.).
This section describes an intercomparison study between ROS-Online and the offline methods implemented at IGE (Calas et al., 2017; Dominutti et al., 2025). The initial concentration of the reagents (AA, DTT, TNB) can significantly influence OP measurement (Lin and Yu, 2011), requiring us to adapt the concentrations of the offline method compared to the routine conditions to enable a comprehensive comparison of ROS-Online and offline OP techniques. Therefore, same samples and identical initial antioxidant quantity of matter, or moles' number n ( = 2.40 × 10−9 mol; = 1.25 × 10−9 mol) were used in the respective measurement cells, i.e. flow cells or plate reader wells for online and offline methods, respectively. The calculation of absorbance has also been adjusted to these new conditions. For these experiments, the ROS-Online samples (control and PM samples) were introduced directly in the liquid form into the BioSampler® without pumping atmospheric air. The comparison between online and offline methods was made by analysing in triplicate the samples prepared and already described in Sect. 4. The three replicates used to calculate the standard deviations for the online method were performed within the same day. For the offline measurements, standard deviations were calculated using the 6 × 3 replicates = 18 readings of the TECAN microplate reader.
Figure 6a–c show the results of the intercomparison (no blank correction) for the positive control solutions of CuCl2 and PQN as analysed by ROS-Online and the offline IGE method. These plots demonstrated the excellent concordance between the 2 methods with linear regressions having R2 > 0.99 and slopes (s) close to 1 (s = 0.99 for all three positive controls).
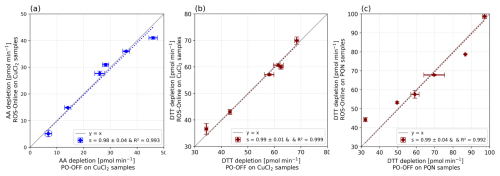
Figure 6Comparison of offline and online methods using: (a, b) CuCl2 samples for AA and DTT assays, respectively and (c) PQN samples on DTT assay. Each sample was analysed in triplicate, with both methods. The vertical error bar represents ± 1σ and the horizontal error bar represents the TECAN microplate variability of 18 measurements (6 readings for each triplicate of each range point). Grey lines represent the line y = x.
Figure 7 shows the results of ROS-Online vs. offline analyses with AA and DTT assays when exposed to PM10 concentrations in the range [0.05–1.05] and [0.05–1.86] µg PM mL−1 for AA and DTT assays, respectively. Quite large error bars on the TECAN microplate reader when measuring AA depletion can be noticed. It should be noted that the offline measurements presented here were performed under the same operational conditions as the online method to allow direct comparison, which may have affected the typical repeatability of the offline protocol. Despite this, the significantly lower variability observed in the online data highlights the improved repeatability and robustness of the ROS-Online instrument. Correlations coefficients of R2 > 0.99 with s values close to 1 (s = 0.95 ± 0.04 and s = 1.01 ± 0.01, for AA and DTT assays, respectively) show again a very similar response between the two techniques with PM extracts. These results are encouraging in the way of providing measurements comparable to the offline methods, themselves currently under harmonisation and standardisation (Dominutti et al., 2025) to comply with response to the new European Directive on air quality.
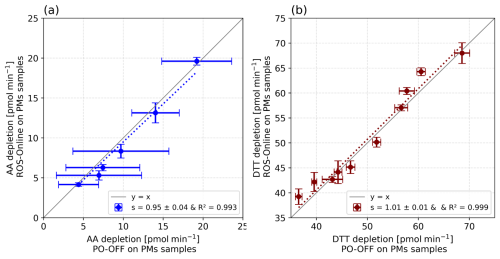
Figure 7Comparison of offline and online methods using: (a) Bern traffic site filter for AA assay, and (b) Grenoble urban background winter filter for DTT assay. Each sample was analysed in triplicate with both methods. The vertical error bar represents ± 1σ and the horizontal error bar represents the TECAN microplate variability of 18 measurements (6 readings for each triplicate of each range point). Grey lines represent the line y = x.
6.1 Site sampling and meteorological conditions
The ROS-Online device was deployed from 1 to 15 September 2023 at the air quality monitoring station “Les Bossons” from the air quality monitoring network in Auvergne – Rhône-Alpes. This site is located in the Arve Valley (45°54′26′′ N; 6°50′45; 1049 m a.s.l. – https://www.atmo-auvergnerhonealpes.fr/dataviz/mesures-aux-stations, last access: 24 November 2025). It is subjected to frequent pollution episodes, particularly in winter due to temperature inversions and intense domestic biomass burning (Quimbayo-Duarte et al., 2021; Weber et al., 2018). This traffic site is located on the roadside of the “Route blanche” with an average of 6300 vehicles (with a large share of trucks) every day during the sampling campaign. The instruments were sheltered in an enclosed cabin with a controlled temperature set at T = 22 ± 4 °C. The station houses a continuous ambient air monitor 1405-F TEOM™ for PM10 mass concentration measurements, and a NOx analyser (model APNA-370, HORIBA). Additional atmospheric observations for PM10 mass concentration (ambient air monitor 1405-F TEOM™) and O3 (ambient ozone monitor – model APOA-370 - HORIBA) were also obtained from the air quality monitoring station “Chamonix” (45°55′21′′ N; 6°52′512; 1038 m a.s.l.), located in Chamonix's city centre, about 2 km away. Meteorological data were obtained from the “Passy” station, located 13 km west on a lower part of the Arve Valley (https://www.infoclimat.fr/opendata/, last access: 24 November 2025) and are presented in the Supplement (Fig. S3). A rainfall episode began during the night of 13 to 14 September and lasted until the end of the campaign (15 September 2023).
6.2 ROS-Online measurements set-up
To maximise the PMs collection, the instrument was set for a sample duration of 30 min at a flowrate of 10.5 L min−1. A protective grid was placed on the inlet line in order to protect the instrument from larger particles that might have clogged the BioSampler® or damaged the FCs. The prototype operated continuously without technical failure during the 15 d campaign. Due to a focus on the AA assay, more likely to react to a traffic source than the DTT assay, OPDTT measurements were maintained from 1 to 6 September only, allowing a higher resolution of OPAA measurements after this date. Indeed, a complete measurement cycle, with both AA and DTT lines operating in parallel, is constrained by the DTT assay, which requires 15 min for measurement – compared to 10 min for the AA assay – as well as additional time for titration point preparation and rinsing. Disabling the DTT line to perform AA-only measurements eliminates this dormant time, thereby enabling higher temporal resolution for the ROS-Online system. Blanks for both assays were very stable: mean blank depletion value for the AA assay was = (0.58 ± 0.24) pmol min−1 (342 occurrences, 15 d) and let us calculate a LOD of (1.30 ± 0.24) pmol min−1. For the DTT assay, = (25.4 ± 4.0) pmol min−1 (over 118 occurrences, 6 d) and LOD = (37.3 ± 4.0) pmol min−1. Finally, evaporation during sampling was evaluated to be (1.3 ± 0.1) mL with minimum and maximum values of 0.8 and 1.9 mL, respectively, mainly depending on the temperature difference between the outside and the shelter's thermostated temperature. The reported evaporation of approximately 1.3 mL corresponds to the average loss measured during each 30 min sampling period and is systematically corrected for during each analytical cycle. The BioSampler® was filled with 20 mL of MQ water before each cycle.
6.3 Results
The weather conditions during the campaign were very stable from 1 to 13 September, with anticyclonic conditions leading to similar daily cycles of temperature, wind and humidity (Fig. S3). Figure 8 shows the average daily cycles of OP measurements, taking into account the full data series for OPAA (1st to 13th) and the overall available dataset of OPDTT (1st to 6th).

Figure 8Average diurnal cycle of 13 d of OPAA measurements (left) and of 6 d of OPDTT measurements (right) observed at Les Bossons [pmol min−1 m−3]. Dashed coloured lines show the averaged diurnal variation for all available measurements, grey and black lines the 3 and 6 h rolling means. Confidence intervals correspond to ± 1σ.
Diurnal cycles of both OPAA and OPDTT measurements do not show the same variations, reflecting the different sensitivity to the aerosol composition of these two probes. OPAA seems to stay at background level (40 pmol min−1 m−3) at night and then rises up around 09:00 until 16:00 LT, reaching twice its background value with a peak of ∼ 70 pmol min−1 m−3, before decreasing at a similar rate than its increase reaching background level again around midnight. The OPDTT signal is less structured with OP values ∼ 5 times that of OPAA at background value (midnight). OPDTT signal exhibits 3 main peaks at ∼ 03:00, noon and ∼ 18:00, reaching ∼ 300 pmol min−1 m−3 for the latter peak. The signal drops rapidly to its background level from 21:00 onwards.
As shown if Fig. 9a, b, d and e, NOx and O3 diurnal cycles show common behaviours for urban traffic sites with maximum levels of NOx reached in the morning (Fig. 9b and e) and maximum ozone concentration in late afternoon (Fig. 9a and d), according to the well-established photochemical equilibrium relationship (Leighton, 1961). OPAA signal is following the atmospheric photochemistry with a strong correlation with the evolution of the ozone concentration, both curves peaking in late afternoon (Fig. 9a). While our laboratory experiments showed that ozone had only a limited effect on the OPAA (Sect. 3.2.1), this covariation is probably the result of the rising of PM concentrations in the afternoon (Fig. 9c) due to primary but more likely secondary emissions as well as boundary layer evolutions. Oxidation (aging) of PM in the afternoon, as shown with AMS measurement at a nearby site during the DECOMBIO project (Jaffrezo et al., 2018), may explain the delay between OPAA and PM peaks on Fig. 9c.
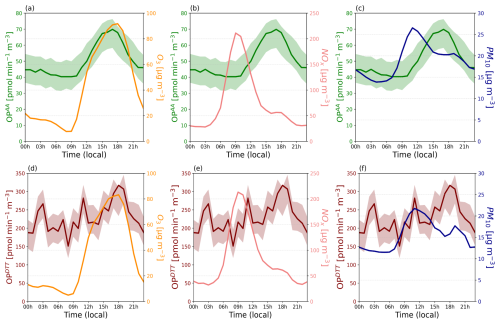
Figure 9Diurnal O3 [µg m−3] (a, d) observed at the measuring sites Chamonix; NOx [µg m−3] (b, e), and PM10 [ µg m−3] (c, f), observed at the measuring sites Les Bossons. Green lines on panels (a), (b), and (d) represents OPAA [pmol min−1 m−3] and brown lines on panels (d), (e), and (f) OPDTT [pmol min−1 m−3]. The 3h rolling mean datasets are used for each parameter. Confidence intervals correspond to ± 1σ.
The general behaviour of OPDTT signal is similar to that of OPAA, especially, OPDTT higher peak corresponds to the O3 peak (Fig. 9d), which could again be explain by the oxidation of PM (Fig. 9f). However, AA and DTT assays provide different information: the sharper OPDTT signal, with 2 narrow peaks observed at around 03:00 and 12:00, may illustrate a local influence from sources or processes to be identified. Finally, during the rainy episode (13 to 15 September) a resulting loss of PM10 and OPAA diurnal cyclicity was observed, probably due to PM scavenging and/or perturbation of a change in boundary layer dynamics. Nevertheless, the OPAA signal increased, still following the O3 daily maximum levels during those 3 rainy days (see Sect. 2.1). Overall, this short field study with the ROS-Online device shows that it is capable to perform dual time series of OP measurement with hourly measurements. These measurements indicate that OP can vary by nearly a factor of two within a day, due to a complex interplay between emissions, aging, and atmospheric dynamics.
The ROS-Online device represents a significant leap forward in measuring the oxidative potential (OP) of ambient particulate matter and soluble gases. Its innovative design, incorporating dual independent lines for simultaneous OPAA and OPDTT assays, allows for near-real-time measurements with high reliability and specificity. Notably, ROS-Online has the potential to capture oxidative potential contributions from both particulate matter and soluble gaseous species present in the BioSampler® solution, providing a more comprehensive assessment of air quality compared to the traditional offline methods that collect PM only. However, current results on gaseous OP are preliminary, and further controlled experiments are required to fully characterize the instrument's response to atmospheric gases. The high linearity and low limits of detection (0.58 ± 0.24) pmol min−1 for AA and (25.4 ± 4.0) pmol min−1 for DTT, underscore its superior performance relative to existing prototypes. The ROS-Online device underwent controlled experiments and field tests to evaluate its performance and versatility.
The experiments in Sect. 3 provided insights into the oxidative potential (OP) responses of two assays, OPAA and OPDTT, when exposed to different aerosol compositions and concentrations. The OPAA assay confirmed its strong sensitivity to metals, particularly CuCl2, showing a proportional decrease in OP with decreasing PM concentration, while OPDTT appeared to reach a saturation point, suggesting a possible reaction plateau. For naphthoquinone (NQ), OP values increased with PM concentration, though not proportionally, likely due to complex aerosol generation dynamics and surface interactions. These findings highlight the different reactivity mechanisms of the two assays towards inorganic and organic aerosols.
Calibration using CuCl2 and 9,10-phenanthrenequinone (PQN) demonstrated acceptable linearity for environmental applications for both OPAA and OPDTT assays, highlighting its sensitivity to both inorganic and organic classes of atmospheric oxidants. Intercomparison with established offline method confirmed efficiency, with high correlation coefficients (R2 > 0.90) for both assays on a large range of concentrations, which is very encouraging for future deployment of ROS-Online. However, in order to associate offline and online measurements, thorough investigations need to be carried out to determine particle trapping between the two methods, exposure times, intrinsic OP values at different sites subjected to different atmospheres, etc.
Field deployment at an urban background site over a 15 d period provided continuous, near-real-time measurements of OPAA and OPDTT. The data revealed distinct diurnal patterns, with OPAA peaking in the afternoon, correlating with photochemical activity and pollutant levels (O3, and PM10). OPDTT measurements showed analogous, but not equivalent trends, with additional peaks indicating sensitivity to localized sources. The device's stability and consistent performance throughout the campaign underscore its suitability for long-term air quality monitoring.
Potential interference from brown carbon (BrC) in our absorbance-based measurements. BrC is known to absorb light in the UV-visible range, and therefore may contribute to the signal at the wavelengths used in our study (265 and 412 nm). As shown by Wu et al. (2023), BrC can contribute significantly to light absorption during biomass burning events, particularly at shorter wavelengths, with its absorption decreasing toward the visible range. Similarly, Basnet et al. (2024) demonstrated that emissions from residential biomass combustion in Europe contain BrC with strong absorption between 300 and 500 nm, which could influence absorbance measurements if not properly accounted for. In our method, absorbance is used to monitor the kinetic consumption or production of reagents, not just the absolute absorbance of the solution. Furthermore, we systematically include blanks and controls to correct for background absorbance, including that potentially attributable to BrC. This approach minimizes the influence of static absorbance contributions from sample matrix components.
A key strength of ROS-Online is its versatility across diverse environmental conditions. The possibility to adapt antioxidant concentrations allows OP measurements in both relatively clean European urban environments and highly polluted regions and or industrial sites, where particulate matter concentrations are significantly higher. To improve sensitivity in low-PM environments, i.e. < 20 µg m−3, ROS-Online can operate with modified reagent concentrations. Any such adjustments require calibration and documentation to ensure comparability. This flexibility ensures accurate assessments regardless of pollution levels, making the device suitable for global deployment.
The device's ability to operate continuously to varying pollution levels reinforces its potential for integration into air quality networks worldwide. Its short temporal resolution (∼ 1 h) enables dynamic tracking of fast-changing atmospheric conditions, essential for understanding pollutant health impacts. Moreover, its sturdiness, demonstrated by continuous operation in field tests, highlights its potential for deployment in urban environments, industrial sites, and high-pollution areas.
ROS-Online's ability to interface with other online chemical analysers positions it as a valuable tool for source apportionment and health risk assessment, supporting regulatory efforts and advancing public health research. These innovations open new pathways for integrating OP measurements into routine air quality monitoring. The device not only meets the growing demand for precise, real-time air quality data but also aligns with the European directive on air quality, emphasizing the relevance of oxidative potential in health studies. Its deployment across air quality stations could be pivotal for pollution monitoring, paving the way for more targeted mitigation strategies and better public health status.
The data are available upon request.
The supplement related to this article is available online at https://doi.org/10.5194/amt-18-7085-2025-supplement.
GU, and JLJ funded the project. The ROS-Online instrument was designed and developed by GF, LR, LP and AB under the supervision of GU and JLJ. LR, GF and LP developed the LabVIEW instrument control software, with the coordination of AB. JC, MB, TM and AM carried out laboratory tests and analyses for the calibration and intercomparison of online and offline methods under the supervision of AB and GU. AB and BG designed, established and carried out the tests in the vein. Finally, AB designed, established and carried out the ozone tests. AB, BG, SH, JLJ and GU wrote the manuscript. All co-authors reviewed the manuscript.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. While Copernicus Publications makes every effort to include appropriate place names, the final responsibility lies with the authors. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
The authors greatly thank the analytical staff of the IGE Air-O-Sol platform and the technical staff of the IGE for their help and support. Special thanks are due to Stephan Houdier, whose diligent work on the review process and meticulous English editing greatly enhanced the quality of this paper
The research underpinning these results was supported by funding from UGA grants, including the Innovative Grant (2016) and Labex@2020 Valorisation (2017), as well as the CNRS Pre-Maturation Grant. Additionally, this work was partially funded by SATT Linksium.
This paper was edited by Francis Pope and reviewed by three anonymous referees.
Ayres, J. G., Borm, P., Cassee, F. R., Castranova, V., Donaldson, K., Ghio, A., Harrison, R. M., Hider, R., Kelly, F., Kooter, I. M., Marano, F., Maynard, R. L., Mudway, I., Nel, A., Sioutas, C., Smith, S., Baeza-Squiban, A., Cho, A., Duggan, S., and Froines, J.: Evaluating the Toxicity of Airborne Particulate Matter and Nanoparticles by Measuring Oxidative Stress Potential—A Workshop Report and Consensus Statement, Inhal. Toxicol., 20, 75–99, https://doi.org/10.1080/08958370701665517, 2008.
Basak, S., Shaik, L., and Chakraborty, S.: Effect of ultraviolet and pulsed light treatments on ascorbic acid content in fruit juices-A review of the degradation mechanism, Food Chemistry Advances, 2, 100333, https://doi.org/10.1016/j.focha.2023.100333, 2023.
Basnet, S., Hartikainen, A., Virkkula, A., Yli-Pirilä, P., Kortelainen, M., Suhonen, H., Kilpeläinen, L., Ihalainen, M., Väätäinen, S., Louhisalmi, J., Somero, M., Tissari, J., Jakobi, G., Zimmermann, R., Kilpeläinen, A., and Sippula, O.: Contribution of brown carbon to light absorption in emissions of European residential biomass combustion appliances, Atmos. Chem. Phys., 24, 3197–3215, https://doi.org/10.5194/acp-24-3197-2024, 2024.
Bates, J. T., Weber, R. J., Abrams, J., Verma, V., Fang, T., Klein, M., Strickland, M. J., Sarnat, S. E., Chang, H. H., Mulholland, J. A., Tolbert, P. E., and Russell, A. G.: Reactive Oxygen Species Generation Linked to Sources of Atmospheric Particulate Matter and Cardiorespiratory Effects, Environ. Sci. Technol., 49, 13605–13612, https://doi.org/10.1021/acs.est.5b02967, 2015.
Bates, J. T., Fang, T., Verma, V., Zeng, L., Weber, R. J., Tolbert, P. E., Abrams, J. Y., Sarnat, S. E., Klein, M., Mulholland, J. A., and Russell, A. G.: Review of Acellular Assays of Ambient Particulate Matter Oxidative Potential: Methods and Relationships with Composition, Sources, and Health Effects, Environ. Sci. Technol., 53, 4003–4019, https://doi.org/10.1021/acs.est.8b03430, 2019.
Bellini, E. and De Tullio, M. C.: Ascorbic Acid and Ozone: Novel Perspectives to Explain an Elusive Relationship, Plants (Basel), 8, 122, https://doi.org/10.3390/plants8050122, 2019.
BioSampler: https://www.skcltd.com/products2/bioaerosol- sampling/biosampler.html, last access: 9 July 2024.
Bøifot, K. O., Skogan, G., and Dybwad, M.: Sampling efficiency and nucleic acid stability during long-term sampling with different bioaerosol samplers, Environ. Monit. Assess., 196, 577, https://doi.org/10.1007/s10661-024-12735-7, 2024.
Borlaza, L. J., Weber, S., Marsal, A., Uzu, G., Jacob, V., Besombes, J.-L., Chatain, M., Conil, S., and Jaffrezo, J.-L.: Nine-year trends of PM10 sources and oxidative potential in a rural background site in France, Atmos. Chem. Phys., 22, 8701–8723, https://doi.org/10.5194/acp-22-8701-2022, 2022.
Bresgen, N. and Eckl, P. M.: Oxidative stress and the homeodynamics of iron metabolism, Biomolecules, 5, 808–847, https://doi.org/10.3390/biom5020808, 2015.
Brock, C. A., Froyd, K. D., Dollner, M., Williamson, C. J., Schill, G., Murphy, D. M., Wagner, N. J., Kupc, A., Jimenez, J. L., Campuzano-Jost, P., Nault, B. A., Schroder, J. C., Day, D. A., Price, D. J., Weinzierl, B., Schwarz, J. P., Katich, J. M., Wang, S., Zeng, L., Weber, R., Dibb, J., Scheuer, E., Diskin, G. S., DiGangi, J. P., Bui, T., Dean-Day, J. M., Thompson, C. R., Peischl, J., Ryerson, T. B., Bourgeois, I., Daube, B. C., Commane, R., and Wofsy, S. C.: Ambient aerosol properties in the remote atmosphere from global-scale in situ measurements, Atmos. Chem. Phys., 21, 15023–15063, https://doi.org/10.5194/acp-21-15023-2021, 2021.
Calas, A., Uzu, G., Martins, J. M. F., Voisin, D., Spadini, L., Lacroix, T., and Jaffrezo, J.-L.: The importance of simulated lung fluid (SLF) extractions for a more relevant evaluation of the oxidative potential of particulate matter, Sci. Rep., 7, 11617, https://doi.org/10.1038/s41598-017-11979-3, 2017.
Calas, A., Uzu, G., Kelly, F. J., Houdier, S., Martins, J. M. F., Thomas, F., Molton, F., Charron, A., Dunster, C., Oliete, A., Jacob, V., Besombes, J.-L., Chevrier, F., and Jaffrezo, J.-L.: Comparison between five acellular oxidative potential measurement assays performed with detailed chemistry on PM10 samples from the city of Chamonix (France), Atmos. Chem. Phys., 18, 7863–7875, https://doi.org/10.5194/acp-18-7863-2018, 2018.
Campbell, S. J., Utinger, B., Lienhard, D. M., Paulson, S. E., Shen, J., Griffiths, P. T., Stell, A. C., and Kalberer, M.: Development of a Physiologically Relevant Online Chemical Assay To Quantify Aerosol Oxidative Potential, Anal. Chem., 91, 13088–13095, https://doi.org/10.1021/acs.analchem.9b03282, 2019.
Campbell, S. J., Utinger, B., Barth, A., Paulson, S. E., and Kalberer, M.: Iron and Copper Alter the Oxidative Potential of Secondary Organic Aerosol: Insights from Online Measurements and Model Development, Environ. Sci. Technol., 57, 13546–13558, https://doi.org/10.1021/acs.est.3c01975, 2023.
Campbell, S. J., Barth, A., Chen, G. I., Tremper, A. H., Priestman, M., Ek, D., Gu, S., Kelly, F. J., Kalberer, M., and Green, D. C.: High time resolution quantification of PM2.5 oxidative potential at a Central London roadside supersite, Environ. Int., 193, 109102, https://doi.org/10.1016/j.envint.2024.109102, 2024.
Campbell, S. J., Utinger, B., Barth, A., Leni, Z., Zhang, Z.-H., Resch, J., Li, K., Steimer, S. S., Banach, C., Gfeller, B., Wragg, F. P. H., Westwood, J., Wolfer, K., Bukowiecki, N., Ihalainen, M., Yli-Pirilä, P., Somero, M., Kortelainen, M., Louhisalmi, J., Sklorz, M., Czech, H., di Bucchianico, S., Streibel, T., Delaval, M. N., Ruger, C., Baumlin, N., Salathe, M., Fang, Z., Pardo, M., D'Aronco, S., Giorio, C., Shi, Z., Harrison, R. M., Green, D. C., Kelly, F. J., Rudich, Y., Paulson, S. E., Sippula, O., Zimmermann, R., Geiser, M., and Kalberer, M.: Short-lived reactive components substantially contribute to particulate matter oxidative potential, Science Advances, 11, eadp8100, https://doi.org/10.1126/sciadv.adp8100, 2025.
Carlino, A., Romano, M. P., Lionetto, M. G., Contini, D., and Guascito, M. R.: An Overview of the Automated and On-Line Systems to Assess the Oxidative Potential of Particulate Matter, Atmosphere, 14, 256, https://doi.org/10.3390/atmos14020256, 2023.
Chang, Y.-P., Wu, S.-J., Lin, M.-S., Chiang, C.-Y., and Huang, G. G.: Ionic-strength and pH dependent reactivities of ascorbic acid toward ozone in aqueous micro-droplets studied by aerosol optical tweezers, Phys. Chem. Chem. Phys., 23, 10108–10117, https://doi.org/10.1039/D0CP06493A, 2021.
Charrier, J. G. and Anastasio, C.: On dithiothreitol (DTT) as a measure of oxidative potential for ambient particles: evidence for the importance of soluble transition metals, Atmos. Chem. Phys., 12, 9321–9333, https://doi.org/10.5194/acp-12-9321-2012, 2012.
Chen, L., Gonze, E., Ondarts, M., Outin, J., and Gonthier, Y.: Electrostatic precipitator for fine and ultrafine particle removal from indoor air environments, Sep. Purif. Technol., 247, 116964, https://doi.org/10.1016/j.seppur.2020.116964, 2020.
Cheng, Y.-H. and Lin, Y.-L.: Measurement of Particle Mass Concentrations and Size Distributions in an Underground Station, Aerosol Air Qual. Res., 10, 22–29, https://doi.org/10.4209/aaqr.2009.05.0037, 2010.
Cho, A. K., Sioutas, C., Miguel, A. H., Kumagai, Y., Schmitz, D. A., Singh, M., Eiguren-Fernandez, A., and Froines, J. R.: Redox activity of airborne particulate matter at different sites in the Los Angeles Basin, Environ. Res., 99, 40–47, https://doi.org/10.1016/j.envres.2005.01.003, 2005.
Daellenbach, K. R., Uzu, G., Jiang, J., Cassagnes, L.-E., Leni, Z., Vlachou, A., Stefenelli, G., Canonaco, F., Weber, S., Segers, A., Kuenen, J. J. P., Schaap, M., Favez, O., Albinet, A., Aksoyoglu, S., Dommen, J., Baltensperger, U., Geiser, M., El Haddad, I., Jaffrezo, J.-L., and Prévôt, A. S. H.: Sources of particulate-matter air pollution and its oxidative potential in Europe, Nature, 587, 414–419, https://doi.org/10.1038/s41586-020-2902-8, 2020.
Damodaran, S.: Estimation of disulfide bonds using 2-nitro-5-thiosulfobenzoic acid: Limitations, Anal. Biochem., 145, 200–204, https://doi.org/10.1016/0003-2697(85)90348-3, 1985.
Delfino, R. J., Staimer, N., Tjoa, T., Gillen, D. L., Schauer, J. J., and Shafer, M. M.: Airway inflammation and oxidative potential of air pollutant particles in a pediatric asthma panel, J. Expo. Sci. Environ. Epidemiol., 23, 466–473, https://doi.org/10.1038/jes.2013.25, 2013.
Delgado-Saborit, J. M., Alam, M. S., Godri Pollitt, K. J., Stark, C., and Harrison, R. M.: Analysis of atmospheric concentrations of quinones and polycyclic aromatic hydrocarbons in vapour and particulate phases, Atmos. Environ., 77, 974–982, https://doi.org/10.1016/j.atmosenv.2013.05.080, 2013.
Denier van der Gon, H., Gerlofs-Nijland, M., Gehrig, R., Gustafsson, M., Janssen, N., Harrison, R., Hulskotte, J., Johansson, C., Jozwicka, M., Keuken, M., Krijgsheld, K., Ntziachristos, L., Riediker, M., and Cassee, F.: The Policy Relevance of Wear Emissions from Road Transport, Now and in the FutureAn International Workshop Report and Consensus Statement, Journal of the Air & Waste Management Association (1995), 63, 136–49, https://doi.org/10.1080/10962247.2012.741055, 2013.
Dominutti, P. A., Borlaza, L. J. S., Sauvain, J.-J., Thuy, V. D. N., Houdier, S., Suarez, G., Jaffrezo, J.-L., Tobin, S., Trébuchon, C., Socquet, S., Moussu, E., Mary, G., and Uzu, G.: Source apportionment of oxidative potential depends on the choice of the assay: insights into 5 protocols comparison and implications for mitigation measures, Environ. Sci.: Atmos., 3, 1497–1512, https://doi.org/10.1039/D3EA00007A, 2023.
Dominutti, P. A., Jaffrezo, J.-L., Marsal, A., Mhadhbi, T., Elazzouzi, R., Rak, C., Cavalli, F., Putaud, J.-P., Bougiatioti, A., Mihalopoulos, N., Paraskevopoulou, D., Mudway, I., Nenes, A., Daellenbach, K. R., Banach, C., Campbell, S. J., Cigánková, H., Contini, D., Evans, G., Georgopoulou, M., Ghanem, M., Glencross, D. A., Guascito, M. R., Herrmann, H., Iram, S., Jovanović, M., Jovašević-Stojanović, M., Kalberer, M., Kooter, I. M., Paulson, S. E., Patel, A., Perdrix, E., Pietrogrande, M. C., Mikuška, P., Sauvain, J.-J., Seitanidi, K., Shahpoury, P., Souza, E. J. d. S., Steimer, S., Stevanovic, S., Suarez, G., Subramanian, P. S. G., Utinger, B., van Os, M. F., Verma, V., Wang, X., Weber, R. J., Yang, Y., Querol, X., Hoek, G., Harrison, R. M., and Uzu, G.: An interlaboratory comparison to quantify oxidative potential measurement in aerosol particles: challenges and recommendations for harmonisation, Atmos. Meas. Tech., 18, 177–195, https://doi.org/10.5194/amt-18-177-2025, 2025.
Dovrou, E., Lelieveld, S., Poeschl, U., and Berkemeier, T.: The Contribution of Reactive Gas-phase Pollutants to Oxidative Stress in the Human Lung, GH15A-0609, bibcode: 2021AGUFMGH15A0609D, 2021.
Eiguren-Fernandez, A., Kreisberg, N., and Hering, S.: An online monitor of the oxidative capacity of aerosols (o-MOCA), Atmos. Meas. Tech., 10, 633–644, https://doi.org/10.5194/amt-10-633-2017, 2017.
Essodolom, P., Ekpetsi Chantal, B., Mamatchi, M., and Kousanta, A.: Effect of temperature on the degradation on acid ascorbic acid (vitamin C) contained in infant supplement flours during the preparation of porridges, IJAR, 8, 116–121, https://doi.org/10.21474/IJAR01/10605, 2020.
Eyer, P., Worek, F., Kiderlen, D., Sinko, G., Stuglin, A., Simeon-Rudolf, V., and Reiner, E.: Molar absorption coefficients for the reduced Ellman reagent: reassessment, Anal. Biochem., 312, 224–227, https://doi.org/10.1016/s0003-2697(02)00506-7, 2003.
Fang, T., Verma, V., Guo, H., King, L. E., Edgerton, E. S., and Weber, R. J.: A semi-automated system for quantifying the oxidative potential of ambient particles in aqueous extracts using the dithiothreitol (DTT) assay: results from the Southeastern Center for Air Pollution and Epidemiology (SCAPE), Atmos. Meas. Tech., 8, 471–482, https://doi.org/10.5194/amt-8-471-2015, 2015.
Fang, T., Verma, V., Bates, J. T., Abrams, J., Klein, M., Strickland, M. J., Sarnat, S. E., Chang, H. H., Mulholland, J. A., Tolbert, P. E., Russell, A. G., and Weber, R. J.: Oxidative potential of ambient water-soluble PM2.5 in the southeastern United States: contrasts in sources and health associations between ascorbic acid (AA) and dithiothreitol (DTT) assays, Atmos. Chem. Phys., 16, 3865–3879, https://doi.org/10.5194/acp-16-3865-2016, 2016.
Fuller, S. J., Wragg, F. P. H., Nutter, J., and Kalberer, M.: Comparison of on-line and off-line methods to quantify reactive oxygen species (ROS) in atmospheric aerosols, Atmos. Environ., 92, 97–103, https://doi.org/10.1016/j.atmosenv.2014.04.006, 2014.
Gao, D., Fang, T., Verma, V., Zeng, L., and Weber, R. J.: A method for measuring total aerosol oxidative potential (OP) with the dithiothreitol (DTT) assay and comparisons between an urban and roadside site of water-soluble and total OP, Atmos. Meas. Tech., 10, 2821–2835, https://doi.org/10.5194/amt-10-2821-2017, 2017.
Gillies, J. A., Gertler, A. W., Sagebiel, J. C., and Dippel, W. A.: On-road particulate matter (PM2.5 and PM10) emissions in the Sepulveda Tunnel, Los Angeles, California, Environ. Sci. Technol., 35, 1054–1063, https://doi.org/10.1021/es991320p, 2001.
Godri, K. J., Harrison, R. M., Evans, T., Baker, T., Dunster, C., Mudway, I. S., and Kelly, F. J.: Increased Oxidative Burden Associated with Traffic Component of Ambient Particulate Matter at Roadside and Urban Background Schools Sites in London, PLoS ONE, 6, e21961, https://doi.org/10.1371/journal.pone.0021961, 2011.
Guo, J., Lv, M., Liu, Z., Qin, T., Qiu, H., zhang, L., Lu, J., Hu, L., Yang, W., and Zhou, D.: Comprehensive performance evaluation of six bioaerosol samplers based on an aerosol wind tunnel, Environ. Int., 183, 108402, https://doi.org/10.1016/j.envint.2023.108402, 2024.
Jaffrezo, J.-L., Besombes, J.-L., Marchand, N., Mocnik, G., Brulfert, G., Chevrier, F., Bertrand, A., Jezek, I., and Allard, J.: DEconvolution de la contribution de la COMbustion de la BIOmasse aux particules dans la vallée de l'Arve. Projet DECOMBIO, report, IGE, ADEME, 2018.
Jiang, H., Ahmed, C. M. S., Canchola, A., Chen, J. Y., and Lin, Y.-H.: Use of Dithiothreitol Assay to Evaluate the Oxidative Potential of Atmospheric Aerosols, Atmosphere, 10, 571, https://doi.org/10.3390/atmos10100571, 2019.
Jiang, H., Ahmed, C. M. S., Zhao, Z., Chen, J. Y., Zhang, H., Canchola, A., and Lin, Y.-H.: Role of functional groups in reaction kinetics of dithiothreitol with secondary organic aerosols, Environ. Pollut., 263, 114402, https://doi.org/10.1016/j.envpol.2020.114402, 2020.
Kachur, A. V., Held, K. D., Koch, C. J., and Biaglow, J. E.: Mechanism of Production of Hydroxyl Radicals in the Copper-Catalyzed Oxidation of Dithiothreitol, Radiat. Res., 147, 409, https://doi.org/10.2307/3579496, 1997.
Keuken, M. P., Moerman, M., Voogt, M., Blom, M., Weijers, E. P., Röckmann, T., and Dusek, U.: Source contributions to PM2.5 and PM10 at an urban background and a street location, Atmos. Environ., 71, 26–35, https://doi.org/10.1016/j.atmosenv.2013.01.032, 2013.
King, L. E. and Weber, R. J.: Development and testing of an online method to measure ambient fine particulate reactive oxygen species (ROS) based on the 2',7'-dichlorofluorescin (DCFH) assay, Atmos. Meas. Tech., 6, 1647–1658, https://doi.org/10.5194/amt-6-1647-2013, 2013.
Kumagai, Y., Koide, S., Taguchi, K., Endo, A., Nakai, Y., Yoshikawa, T., and Shimojo, N.: Oxidation of Proximal Protein Sulfhydryls by Phenanthraquinone, a Component of Diesel Exhaust Particles, Chem. Res. Toxicol., 15, 483–489, https://doi.org/10.1021/tx0100993, 2002.
Leighton, P.: Photochemistry of Air Pollution, 1st edn., ISBN 9780323156455, 1961.
Leni, Z., Cassagnes, L. E., Daellenbach, K. R., Haddad, I. E., Vlachou, A., Uzu, G., Prévôt, A. S. H., Jaffrezo, J.-L., Baumlin, N., Salathe, M., Baltensperger, U., Dommen, J., and Geiser, M.: Oxidative stress-induced inflammation in susceptible airways by anthropogenic aerosol, PLOS ONE, 15, e0233425, https://doi.org/10.1371/journal.pone.0233425, 2020.
Li, J., Leavey, A., Wang, Y., O'Neil, C., Wallace, M. A., Burnham, C.-A. D., Boon, A. C., Babcock, H., and Biswas, P.: Comparing the performance of 3 bioaerosol samplers for influenza virus, J. Aerosol. Sci., 115, 133–145, https://doi.org/10.1016/j.jaerosci.2017.08.007, 2018.
Li, Q., Wyatt, A., and Kamens, R. M.: Oxidant generation and toxicity enhancement of aged-diesel exhaust, Atmos. Environ., 43, 1037–1042, https://doi.org/10.1016/j.atmosenv.2008.11.018, 2009.
Lin, H., Chen, Q., Wang, M., and Chang, T.: Oxidation potential and coupling effects of the fractionated components in airborne fine particulate matter, Environ. Res., 213, 113652, https://doi.org/10.1016/j.envres.2022.113652, 2022.
Lin, P. and Yu, J. Z.: Generation of Reactive Oxygen Species Mediated by Humic-like Substances in Atmospheric Aerosols, Environ. Sci. Technol., 45, 10362–10368, https://doi.org/10.1021/es2028229, 2011.
Lin, X.-T., Hsu, N.-Y., Wang, J.-R., Chen, N.-T., Su, H.-J., and Lin, M.-Y.: Development of an efficient viral aerosol collector for higher sampling flow rate, Environ. Sci. Pollut. Res., 25, 3884–3893, https://doi.org/10.1007/s11356-017-0754-z, 2018.
Liu, X., Zhang, X., Wang, T., Jin, B., Wu, L., Lara, R., Monge, M., Reche, C., Jaffrezo, J.-L., Uzu, G., Dominutti, P., Darfeuil, S., Favez, O., Conil, S., Marchand, N., Castillo, S., de la Rosa, J. D., Stuart, G., Eleftheriadis, K., Diapouli, E., Gini, M. I., Nava, S., Alves, C., Wang, X., Xu, Y., Green, D. C., Beddows, D. C. S., Harrison, R. M., Alastuey, A., and Querol, X.: PM10-bound trace elements in pan-European urban atmosphere, Environ. Res., 260, 119630, https://doi.org/10.1016/j.envres.2024.119630, 2024.
Manigrasso, M., Costabile, F., Liberto, L. D., Gobbi, G. P., Gualtieri, M., Zanini, G., and Avino, P.: Size resolved aerosol respiratory doses in a Mediterranean urban area: From PM10 to ultrafine particles, Environ. Int., 141, 105714, https://doi.org/10.1016/j.envint.2020.105714, 2020.
Manisalidis, I., Stavropoulou, E., Stavropoulos, A., and Bezirtzoglou, E.: Environmental and Health Impacts of Air Pollution: A Review, Frontiers in Public Health, 8, 14, https://doi.org/10.3389/fpubh.2020.00014, 2020.
Mescioglu, E., Paytan, A., Mitchell, B. W., and Griffin, D. W.: Efficiency of bioaerosol samplers: a comparison study, Aerobiologia, 37, 447–459, https://doi.org/10.1007/s10453-020-09686-0, 2021.
Møller, P., Danielsen, P. H., Karottki, D. G., Jantzen, K., Roursgaard, M., Klingberg, H., Jensen, D. M., Christophersen, D. V., Hemmingsen, J. G., Cao, Y., and Loft, S.: Oxidative stress and inflammation generated DNA damage by exposure to air pollution particles, Mutat. Res. Rev. Mutat. Res., 762, 133–166, https://doi.org/10.1016/j.mrrev.2014.09.001, 2014.
Møller, P., Scholten, R., Roursgaard, M., and Krais, A.: Inflammation, oxidative stress and genotoxicity responses to biodiesel emissions in cultured mammalian cells and animals, Crit. Rev. Toxicol., 50, 1–19, https://doi.org/10.1080/10408444.2020.1762541, 2020.
Mudway, I. S., Stenfors, N., Duggan, S. T., Roxborough, H., Zielinski, H., Marklund, S. L., Blomberg, A., Frew, A. J., Sandström, T., and Kelly, F. J.: An in vitro and in vivo investigation of the effects of diesel exhaust on human airway lining fluid antioxidants, Arch. Biochem. Biophys., 423, 200–212, https://doi.org/10.1016/j.abb.2003.12.018, 2004.
Park, M., Joo, H. S., Lee, K., Jang, M., Kim, S. D., Kim, I., Borlaza, L. J. S., Lim, H., Shin, H., Chung, K. H., Choi, Y.-H., Park, S. G., Bae, M.-S., Lee, J., Song, H., and Park, K.: Differential toxicities of fine particulate matters from various sources, Sci. Rep., 8, 17007, https://doi.org/10.1038/s41598-018-35398-0, 2018.
Perrone, M. R., Bertoli, I., Romano, S., Russo, M., Rispoli, G., and Pietrogrande, M. C.: PM2.5 and PM10 oxidative potential at a Central Mediterranean Site: Contrasts between dithiothreitol- and ascorbic acid-measured values in relation with particle size and chemical composition, Atmos. Environ., 210, 143–155, https://doi.org/10.1016/j.atmosenv.2019.04.047, 2019.
Pietrogrande, M. C., Romanato, L., and Russo, M.: Synergistic and Antagonistic Effects of Aerosol Components on Its Oxidative Potential as Predictor of Particle Toxicity, Toxics, 10, 196, https://doi.org/10.3390/toxics10040196, 2022.
Puthussery, J. V., Zhang, C., and Verma, V.: Development and field testing of an online instrument for measuring the real-time oxidative potential of ambient particulate matter based on dithiothreitol assay, Atmos. Meas. Tech., 11, 5767–5780, https://doi.org/10.5194/amt-11-5767-2018, 2018.
Qu, J., Li, Y., Zhong, W., Gao, P., and Hu, C.: Recent developments in the role of reactive oxygen species in allergic asthma, J. Thorac. Dis., 9, E32–E43, https://doi.org/10.21037/jtd.2017.01.05, 2017.
Quimbayo-Duarte, J., Chemel, C., Staquet, C., Troude, F., and Arduini, G.: Drivers of severe air pollution events in a deep valley during wintertime: A case study from the Arve river valley, France, Atmos. Environ., 247, 118030, https://doi.org/10.1016/j.atmosenv.2020.118030, 2021.
Rao, L., Zhang, L., Wang, X., Xie, T., Zhou, S., Lu, S., Liu, X., Lu, H., Xiao, K., Wang, W., and Wang, Q.: Oxidative Potential Induced by Ambient Particulate Matters with Acellular Assays: A Review, Processes, 8, 1410, https://doi.org/10.3390/pr8111410, 2020.
Shahpoury, P., Harner, T., Lammel, G., Lelieveld, S., Tong, H., and Wilson, J.: Development of an antioxidant assay to study oxidative potential of airborne particulate matter, Atmos. Meas. Tech., 12, 6529–6539, https://doi.org/10.5194/amt-12-6529-2019, 2019.
Shahpoury, P., Zhang, Z. W., Arangio, A., Celo, V., Dabek-Zlotorzynska, E., Harner, T., and Nenes, A.: The influence of chemical composition, aerosol acidity, and metal dissolution on the oxidative potential of fine particulate matter and redox potential of the lung lining fluid, Environ. Int., 148, 106343, https://doi.org/10.1016/j.envint.2020.106343, 2021.
Shahpoury, P., Lelieveld, S., Johannessen, C., Berkemeier, T., Celo, V., Dabek-Zlotorzynska, E., Harner, T., Lammel, G., and Nenes, A.: Influence of aerosol acidity and organic ligands on transition metal solubility and oxidative potential of fine particulate matter in urban environments, Sci. Total Environ., 906, 167405, https://doi.org/10.1016/j.scitotenv.2023.167405, 2024a.
Shahpoury, P., Lelieveld, S., Srivastava, D., Baccarini, A., Mastin, J., Berkemeier, T., Celo, V., Dabek-Zlotorzynska, E., Harner, T., Lammel, G., and Nenes, A.: Seasonal Changes in the Oxidative Potential of Urban Air Pollutants: The Influence of Emission Sources and Proton- and Ligand-Mediated Dissolution of Transition Metals, ACS EST Air, 1, 1262–1275, https://doi.org/10.1021/acsestair.4c00093, 2024b.
Sherif, E. M. and Park, S.-M.: Effects of 1,4-naphthoquinone on aluminum corrosion in 0.50 M sodium chloride solutions, Electrochim. Acta, 51, 1313–1321, https://doi.org/10.1016/j.electacta.2005.06.018, 2006.
Song, M., Oh, S.-H., Park, C., and Bae, M.-S.: Review: Analytical Procedure for Dithiothreitol-based Oxidative Potential of PM2.5, Asian Journal of Atmospheric Environment, 15, 13, https://doi.org/10.5572/ajae.2021.015, 2021.
State of Global Air Report 2024: State of Global Air: https://www.stateofglobalair.org/resources/report/state-global-air-report-2024, last access: 16 December 2024.
Stevanovic, S.: Oxidative potential of gas phase combustion emissions – An underestimated and potentially harmful component of air pollution from combustion processes, Atmos. Environ., 158, 227–235, 2017.
Strak, M., Janssen, N. A. H., Godri, K. J., Gosens, I., Mudway, I. S., Cassee, F. R., Lebret, E., Kelly, F. J., Harrison, R. M., Brunekreef, B., Steenhof, M., and Hoek, G.: Respiratory Health Effects of Airborne Particulate Matter: The Role of Particle Size, Composition, and Oxidative Potential – The RAPTES Project, Environ. Health Persp., 120, 1183–1189, https://doi.org/10.1289/ehp.1104389, 2012.
Su, W.-C. and Vincent, J. H.: Towards a general semi-empirical model for the aspiration efficiencies of aerosol samplers in perfectly calm air, J. Aerosol Sci., 35, 1119–1134, https://doi.org/10.1016/j.jaerosci.2004.04.003, 2004.
Su, Y., Wang, W., Wang, W., Zhai, L., Shen, X., Xu, J., and Li, Z.: Re-evaluation of BioSampler and its improvement for on-line, time-resolved monitoring of environmental coarse aerosol, Atmos. Environ., 225, 117249, https://doi.org/10.1016/j.atmosenv.2019.117249, 2020.
Turner, M. C., Andersen, Z. J., Baccarelli, A., Diver, W. R., Gapstur, S. M., Pope III, C. A., Prada, D., Samet, J., Thurston, G., and Cohen, A.: Outdoor air pollution and cancer: An overview of the current evidence and public health recommendations, CA: A Cancer Journal for Clinicians, 70, 460–479, https://doi.org/10.3322/caac.21632, 2020.
Utinger, B., Campbell, S. J., Bukowiecki, N., Barth, A., Gfeller, B., Freshwater, R., Rüegg, H.-R., and Kalberer, M.: An automated online field instrument to quantify the oxidative potential of aerosol particles via ascorbic acid oxidation, Atmos. Meas. Tech., 16, 2641–2654, https://doi.org/10.5194/amt-16-2641-2023, 2023.
Uzu, G., Sauvain, J.-J., Baeza-Squiban, A., Riediker, M., Sánchez Sandoval Hohl, M., Val, S., Tack, K., Denys, S., Pradère, P., and Dumat, C.: In vitro Assessment of the Pulmonary Toxicity and Gastric Availability of Lead-Rich Particles from a Lead Recycling Plant, Environ. Sci. Technol., 45, 7888–7895, https://doi.org/10.1021/es200374c, 2011.
Walker, M. D., Vincent, J. C., Benson, L., Stone, C. A., Harris, G., Ambler, R. E., Watts, P., Slatter, T., López-García, M., King, M.-F., Noakes, C. J., and Thomas, R. J.: Effect of Relative Humidity on Transfer of Aerosol-Deposited Artificial and Human Saliva from Surfaces to Artificial Finger-Pads, Viruses, 14, 1048, https://doi.org/10.3390/v14051048, 2022.
Weber, S., Uzu, G., Calas, A., Chevrier, F., Besombes, J.-L., Charron, A., Salameh, D., Ježek, I., Močnik, G., and Jaffrezo, J.-L.: An apportionment method for the oxidative potential of atmospheric particulate matter sources: application to a one-year study in Chamonix, France, Atmos. Chem. Phys., 18, 9617–9629, https://doi.org/10.5194/acp-18-9617-2018, 2018.
Weber, S., Uzu, G., Favez, O., Borlaza, L. J. S., Calas, A., Salameh, D., Chevrier, F., Allard, J., Besombes, J.-L., Albinet, A., Pontet, S., Mesbah, B., Gille, G., Zhang, S., Pallares, C., Leoz-Garziandia, E., and Jaffrezo, J.-L.: Source apportionment of atmospheric PM10 oxidative potential: synthesis of 15 year-round urban datasets in France, Atmos. Chem. Phys., 21, 11353–11378, https://doi.org/10.5194/acp-21-11353-2021, 2021.
WHO: https://www.who.int/news-room/fact-sheets/detail/ambient-(outdoor)-air-quality-and-health, last access: 29 May 2023.
Wittmaack, K.: In Search of the Most Relevant Parameter for Quantifying Lung Inflammatory Response to Nanoparticle Exposure: Particle Number, Surface Area, or What?, Environ. Health Persp., 115, 187–194, https://doi.org/10.1289/ehp.9254, 2007.
Wragg, F. P. H., Fuller, S. J., Freshwater, R., Green, D. C., Kelly, F. J., and Kalberer, M.: An automated online instrument to quantify aerosol-bound reactive oxygen species (ROS) for ambient measurement and health-relevant aerosol studies, Atmos. Meas. Tech., 9, 4891–4900, https://doi.org/10.5194/amt-9-4891-2016, 2016.
Wu, C., He, C., Brown, Z. E., Miljevic, B., Zhang, C., Wang, H., Wang, B., Morawska, L., and Ristovski, Z.: Light absorption properties of black and brown carbon during the prescribed burning season at an urban background site in Brisbane, Australia, Atmos. Environ., 313, 120072, https://doi.org/10.1016/j.atmosenv.2023.120072, 2023.
Yang, A., Jedynska, A., Hellack, B., Kooter, I., Hoek, G., Brunekreef, B., Kuhlbusch, T. A. J., Cassee, F. R., and Janssen, N. A. H.: Measurement of the oxidative potential of PM2.5 and its constituents: The effect of extraction solvent and filter type, Atmos. Environ., 83, 35–42, https://doi.org/10.1016/j.atmosenv.2013.10.049, 2014.
Yang, M., Ahmed, H., Wu, W., Jiang, B., and Jia, Z.: Cytotoxicity of Air Pollutant 9,10-Phenanthrenequinone: Role of Reactive Oxygen Species and Redox Signaling, BioMed Research International, 2018, 1–15, https://doi.org/10.1155/2018/9523968, 2018.
Yu, H., Puthussery, J. V., and Verma, V.: A semi-automated multi-endpoint reactive oxygen species activity analyzer (SAMERA) for measuring the oxidative potential of ambient PM2.5 aqueous extracts, Aerosol Science and Technology, 54, 304–320, https://doi.org/10.1080/02786826.2019.1693492, 2020.
- Abstract
- Introduction
- The ROS-Online device
- ROS-Online response under semi-controlled environment
- ROS-Online linearity assessment with positive controls and exposed samples
- Inter-comparison of online and offline measurements
- Real-life case study: daily variation of OPAA near a major roadway
- Conclusions and perspectives
- Data availability
- Author contributions
- Competing interests
- Disclaimer
- Acknowledgements
- Financial support
- Review statement
- References
- Supplement
- Abstract
- Introduction
- The ROS-Online device
- ROS-Online response under semi-controlled environment
- ROS-Online linearity assessment with positive controls and exposed samples
- Inter-comparison of online and offline measurements
- Real-life case study: daily variation of OPAA near a major roadway
- Conclusions and perspectives
- Data availability
- Author contributions
- Competing interests
- Disclaimer
- Acknowledgements
- Financial support
- Review statement
- References
- Supplement