the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Evaluation of calibration strategies for accurate δ13CH4 measurements in dry and humid air
Ji Li
Xuguang Chi
Aijun Ding
Weimin Ju
Yongguang Zhang
Jing M. Chen
Accurate determination of the methane isotopic composition (δ13CH4) is essential for attributing emission sources of methane (CH4). However, for measurements with optical instruments, spectral interference from water vapor and instrumental drift often introduce substantial biases in δ13CH4 measurements, particularly for humid air measurements. Although multiple calibration strategies exist, a systematic evaluation of their performance under diverse field conditions remains lacking. Here, we evaluate two calibration strategies for a cavity ring-down spectrometer: a delta-based calibration for δ13CH4 and an isotopologue-specific calibration for 12CH4 and 13CH4. We performed laboratory experiments over a water vapor range of 0.15 %–4.0 % to establish empirical correction functions, quadratic for 12CH4 and 13CH4, and linear for δ13CH4, to remove humidity-induced biases. These correction functions were then applied to field measurements in both dried air at the SORPES stie and humid air at the Jurong site. At the SORPES site where air samples were dried using a Nafion™ dryer, the mean difference in δ13CH4 between the two strategies was ∼0.29 ‰. In contrast, for humid air at the Jurong site, significant inter-method difference (Δδ13CH4) was observed, with which exhibiting a strong correlation with , indicating non-linear spectral effects are most pronounced at lower CH4 concentrations and compromise the performance of delta-based calibration. Notably, only the isotopologue-specific calibration, coupled with an explicit water vapor correction, delivered stable and accurate δ13CH4 measurements across all conditions. This work underscores the need for robust calibration strategies to minimize bias in CH4 isotopic composition measurements.
- Article
(7173 KB) - Full-text XML
- BibTeX
- EndNote
Methane (CH4) is a potent greenhouse gas that plays a key role in climate change, contributing approximately 16.4 % of total anthropogenic radiative forcing (Patra and Khatri, 2022). Its global warming potential is about 28 times greater than that of carbon dioxide (CO2) over a 100-year time horizon, making CH4 a critical target for near-term climate mitigation (Forster et al., 2021; Nisbet et al., 2020; Van Dingenen et al., 2018; Shindell et al., 2012; Ipcc, 2007). The primary sources of CH4 emissions are direct anthropogenic activities (e.g., agriculture, waste, fossil fuels, and biomass burning), and natural and indirect anthropogenic sources, including wetlands, inland waters, and geological seepage. In contrast, its removal from the atmosphere is mainly governed by oxidation with hydroxyl radicals (OH) (Kirschke et al., 2013; Olivier and Berdowski, 2021; Saunois et al., 2020). However, substantial uncertainties remain in these estimates: 20 %–35 % for anthropogenic sources, ∼50 % for wetlands and biomass burning, up to 100 % for inland waters and geological sources, and 10 %–20 % for the OH sink (Saunois et al., 2025). To constrain budgets and design effective reduction strategies, it is essential to distinguish between its diverse emission sources.
The carbon isotopic composition of CH4 (δ13CH4) provides valuable constraints on tracking emission sources (Nisbet et al., 2016; Rice et al., 2016; Schaefer et al., 2016), as microbial, thermogenic, and pyrogenic origins exhibit distinct isotopic signatures (Levin et al., 1993; Bakkaloglu et al., 2022; Ehleringer and Osmond, 1989). δ13CH4-based analysis enables classification of emission types and supports quantitative estimation of CH4 emissions on regional to global scales (De Groot, 2004; Saunois et al., 2020; Lan et al., 2021). However, these applications critically depend on high-precision isotopic measurements, since even small observational biases can propagate into large errors in inferred source signatures (Iaea, 2024; Defratyka et al., 2025; France et al., 2022).
Conventionally, δ13CH4 has been measured using isotope ratio mass spectrometry (IRMS), which provides high precision measurements (≤0.1 ‰) but requires labor-intensive sampling and lacks continuous coverage (Miller et al., 2002; Schaefer et al., 2006; Röckmann et al., 2016). Recent advances in laser-based spectroscopy, particularly cavity ring-down spectroscopy (CRDS) and quantum cascade laser absorption spectroscopy (QCLAS), have enabled in situ and automated δ13CH4 monitoring (Rella et al., 2015; Tuzson et al., 2008). While these techniques offer advantages over conventional IRMS, their measurement accuracy is challenged by spectroscopic interferences, such as water vapor, and by the choice of calibration strategies. This is true whether δ13CH4 is adjusted directly or is derived from isotopologue-specific calibrations, as highlighted in subsequent evaluations (Griffith, 2018; Hoheisel et al., 2019; Saboya et al., 2022).
Water vapor affects measured CH4 mole fractions through dilution and spectral interference, thereby introducing systematic biases in isotopologue-based measurements (Chen et al., 2010; Hoheisel et al., 2019; Saboya et al., 2022). Consequently, whether the sampled air is dried prior to analysis critically affects the accuracy of δ13CH4 measurements, a factor that becomes particularly important in humid environments. In addition, two primary calibration strategies have been developed to retrieve δ13CH4 from laser-based spectroscopic observations (Wen et al., 2013; Griffith, 2018; Griffith et al., 2012; Flores et al., 2017; Tans et al., 2017). One is a direct correction of δ13CH4 (hereafter, delta-based correction), which is relatively straightforward but may retain residual artifacts linked to CH4 concentration (Wen et al., 2013; Pang et al., 2016; Griffith et al., 2012; Braden-Behrens et al., 2017; Flores et al., 2017). Another one is isotopologue-specific correction for 12CH4 and 13CH4 (hereafter, isotopologue-specific correction), in which δ13CH4 is derived from independently corrected isotopologue concentrations, thereby reducing concentration-dependent biases (Griffith, 2018; Wen et al., 2013).
As water vapor remains a dominant limitation for accurate δ13CH4 measurement, physical drying to approximately <0.1 % H2O (dew point °C) is generally recommended to obtain high-precision δ13CH4 measurements at the sub-per-mil level (Rella et al., 2015). Laboratory studies demonstrated a quadratic CH4–H2O relationship, indicating that water vapor affects CH4 through both dilution and spectral interference (Chen et al., 2010; Rella et al., 2013), and a cross-sensitivity of ∼0.54 ‰ per 1 % H2O within 0.15 %–1.5 % H2O (Hoheisel et al., 2019). In comparison, Saboya et al. (2022) applied a linear δ13CH4 correction over 0 %–2.2 % H2O and found CH4 mole fractions to be unaffected by water vapor within this range, while Chen et al. (2010) reported a clear quadratic dependence across 0.6 %–6 % H2O. Moreover, explicit correction functions for the individual isotopologues (12CH4 and 13CH4) remain lacking, and the δ13CH4 corrections vary widely in form, highlighting the need for more robust approaches. Both calibration strategies ideally rely on multi-point calibration using reference gases that span the targeted range, either in mole fractions of 12CH4 and 13CH4 (for isotopologue-specific calibration) or in δ13CH4 (for delta-based calibration) (Wen et al., 2013; Griffith, 2018), but practical limitations persist. Delta-based correction is constrained by scarce isotopic standards (Griffith, 2018) and prone to concentration-dependent biases (Wen et al., 2013; Griffith, 2018), while isotopologue-specific correction can reduce such concentration dependence but lacks well-established water correction functions for 12CH4 and 13CH4. These methodological gaps are particularly critical in humid environments, where water vapor effects are often large.
In this study, we aim to evaluate isotopologue-specific and delta-based calibration strategies for obtaining accurate δ13CH4 measurements for both dry and humid air. To achieve this, we conducted water vapor laboratory experiments to derive empirical correction functions for 12CH4, 13CH4, and δ13CH4. These corrections were then applied to field measurements to assess the accuracy of both calibration strategies for measurements in both dry and humid air.
2.1 The δ13CH4 analyzer
The δ13C-CH4 analyzer used in this study was a G2201-i instrument manufactured by Picarro Inc. (Santa Clara, CA, USA). All laboratory experiments and field measurements reported here were performed using the same G2201-i analyzer, although the field measurements were performed during different campaigns in different years (2018, 2022, and 2025). This instrument is based on the cavity ring-down spectroscopy (CRDS) technique, which enables real-time measurements of CH4 and CO2 isotopologues (12CH4, 13CH4, 12CO2, 13CO2). CRDS quantifies gas absorption by measuring the exponential decay rate of light intensity in a high-reflectivity optical cavity, providing high sensitivity and strong resistance to external interference (Crosson, 2008; Berden and Engeln, 2009; Crosson et al., 2002; Wahl et al., 2006). Here, we evaluate observations of both CH4 mole fractions and δ13CH4 values derived from 12CH4 and 13CH4 measurements.
The instrument is equipped with a dual-laser module that allows automatic switching between CH4 and CO2 isotopologue measurements. During experiments covering a humidity range of 0 %–4.0 %, the cavity temperature and pressure remained highly stable (45.000±0.001 °C and 197.317±0.084 hPa, respectively). The analyzer applies an internal, empirically parameterized water-vapor interference correction based on the measured H2O absorption signal (peak75), which is non-linear and includes a quadratic term, as described in the G2201-i user manual. The analyzer includes an internal water vapor correction algorithm that compensates for humidity effects for the calculation of δ13CH4. According to the manufacturer's white paper, this correction is internally applied to the 12CH4 signal before computing δ13CH4, using an empirically derived function to adjust for H2O-induced spectral interference (Rella, 2012). In this study, all water-vapor correction functions were derived from the raw (uncorrected) outputs. All subsequent analyses were performed using raw 12CH4, 13CH4, and δ13CH4 values corrected offline with the empirical water-vapor correction functions developed here. Manufacturer-corrected outputs were not used in the calibration workflow to avoid potential double correction. As a result, δ13CH4 values are indirectly adjusted for humidity, whereas the raw 12CH4 and 13CH4 mole fractions remain uncorrected and represent humid-air concentrations.
2.2 Experimental design and measurement system
2.2.1 Water vapor correction
To quantify and correct for the influence of water vapor on the δ13CH4 measurements, we performed a water vapor laboratory experiment using a Picarro G2201-i analyzer (Fig. 1a), following a modified setup from Rella et al. (2013). A cotton filter soaked with deionized water was placed at the inlet to gradually humidify the sample stream, forming an artificially humidified phase to derive empirical water vapor correction functions for 12CH4, 13CH4, and δ13CH4. The analyzer continuously sampled a reference gas (Ref0, Table 1) at a flow rate of ∼45 mL min−1 for approximately six hours, during which water vapor mole fraction decreased from ∼4.0 % to ∼0.1 %. Over the entire experiment, the instrument drift between the two dry-reference periods was 1.8 ppb (0.092 %) for 12CH4, 0.02 ppb (0.095 %) for 13CH4, and 0.262 ‰ for δ13CH4 (Fig. 2). Empirical relationships between measured signals and H2O concentration were then derived. For 12CH4 and 13CH4, second-order polynomial fits were applied (Chen et al., 2010; Rella et al., 2013), while δ13CH4 was corrected using a linear regression (Hoheisel et al., 2019). These correction functions were subsequently applied to both reference and field measurements to remove H2O-induced spectral interference.
The main humidity-response experiment was conducted once in this study. As no repeated humidity tests were performed over time, we cannot assess the temporal stability and reproducibility of the fitted correction functions. For long-term applications, periodic validation and, if necessary, re-derivation of the correction functions are recommended.
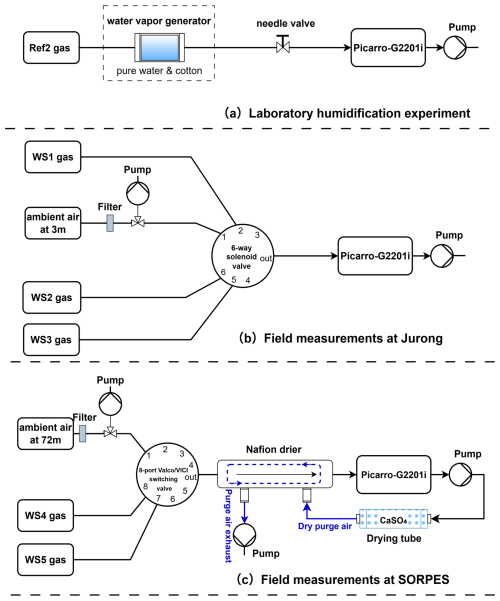
Figure 1Laboratory and field setups for CH4 and δ13 CH4 measurements using a Picarro G2201-i analyzer. (a) Laboratory setup for deriving the water vapor correction function using humidified Ref2 gas. (b) Field setup at the Jurong site using WS1–WS3 and ambient air at 3 m above the ground, streams selected via a solenoid valve. (c) Field setup at the SORPES site with WS4–WS5 and ambient air sampled at 72 m above the ground, equipped with a Nafion™ dryer. Note that a 6-way solenoid valve was used at Jurong, whereas an 8-port Valco or VICI switching valve was used at SORPES.
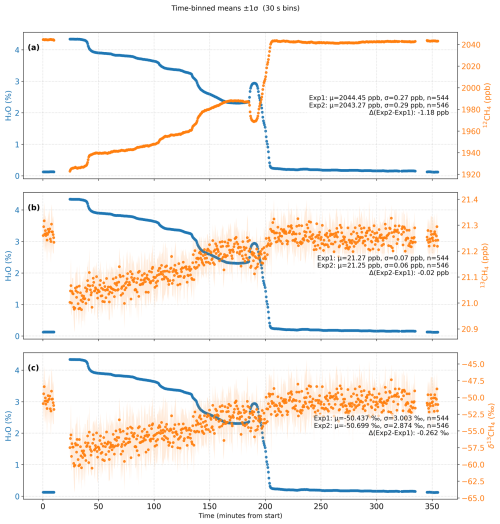
Figure 2Time series of H2O, 12CH4, 13CH4, and δ13CH4 during the controlled humidity experiment. Each point represents a 30 s mean value, and the shaded areas indicate the 1σ standard deviation of each bin. Exp1 and Exp2 correspond to the dry reference gas measurements performed before and after the artificially humidified period, respectively. The annotated statistics in each panel show the mean (μ), standard deviation (σ), number of averaged points (n), and the drift (ΔExp2–Exp1), demonstrating the measurement stability of 12CH4, 13CH4, and δ13CH4 across the humidified period. H2O represents the water vapor mole fraction (in %).
2.2.2 Field observations
The δ13CH4 measurement system was deployed at two sites in the Yangtze River Delta (Fig. 1b, c; Table 2). The Station for Observing Regional Processes of the Earth System (SORPES) station, located on the Xianlin campus of Nanjing University (118°57′10′′ E, 32°07′14′′ N; ∼40 m a.s.l.), represents a regional background site influenced by large-scale anthropogenic emissions (Ding et al., 2016). The Jurong station (31°48′24.59′′N, 119°13′2.15′′E; ∼15 m a.s.l.) is situated in an irrigated rice paddy and characterizes an agricultural ecosystem under a subtropical monsoon climate (Dai et al., 2019; Li et al., 2020).
At both sites, ambient CH4 and δ13CH4 were recorded at 0.5 Hz and aggregated to 5-min means. Reference gases were introduced every six hours to ensure accuracy while conserving calibration gases. Humidity conditions contrasted strongly between two sites: SORPES used a Nafion™ membrane dryers (Perma Pure, USA) to maintain stable H2O (0.04 %–0.40 %), whereas Jurong operated without drying, resulting in elevated H2O (0.93 %–3.5 %) consistent with the paddy environment. Calibration approaches also differed. Table 1 describes the compositions and assigned true values of the reference gases (Ref1–Ref3) and working standard gases (WS1–WS5). At Jurong, ambient measurements of CH4 mole fractions were calibrated using a two-point linear correction based on WS1 and WS3. δ13CH4 was calibrated using the same two reference gases via the linear delta-based calibration (Eq. 8). WS1 and WS3 span a wide CH4 range, but exhibit a relatively narrow δ13CH4 range (Table 1). At SORPES, WS4 and WS5 were used to determine the calibration slope applied throughout the observation period, whereas WS5 served as a single-point reference to correct instrument drift.
Table 1Certified values of CH4 molar fraction and δ13CH4 for dry and humidified reference gases.

Note: CH4 and δ13CH4 are certified values for each standard gas. 13r denotes the ratio between 13CH4 and 12CH4, calculated from Eq. (5). Rsum is the total isotopologue normalising factor for methane, defined in Eq. (6). This correction accounts for the absorption effects introduced by the presence of hydrogen isotopologues such as CH3D in atmospheric methane, in addition to carbon isotopologues. 12CH4 and 13CH4 were derived quantities calculated from assigned total CH4 and δ13CH4; their uncertainties were obtained by uncertainty propagation. In this cylinder set, WS1–WS5 are working standards calibrated against Ref1–Ref3. Ref1–Ref3 have manufacturer-certified CH4 and δ13CH4 values and are used as the reference standards for the calibration of WS1–WS5.
2.2.3 Reference gas measurements
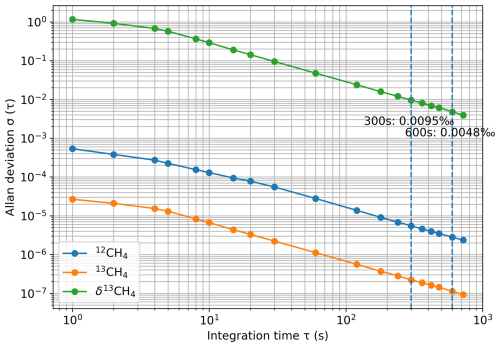
Reference gases were measured in the laboratory and at both field sites to evaluate the performance of the calibration strategies and to ensure consistency of ambient air measurements (Fig. 1b–c). In the laboratory, three reference gases (Ref1–Ref3) were analyzed for 30 min each, while at the Jurong and SORPES sites, three (WS1–WS3) and two (WS4–WS5) reference gases were measured, respectively. Each field measurement lasted 10 min, and the final 5 min were averaged for analysis. This window was chosen to avoid stabilization effects immediately after valve switching and to represent the stable plateau of each injection. The Allan deviation of δ13CH4 (raw) decreases monotonically over the accessible τ range, with σ (300 s) = 0.0095 ‰ and σ (600 s) = 0.0048 ‰ (Fig. A2), and no drift-dominated upturn is observed within this range. Although 600 s yields lower random noise, we retain 5-min averaging as a practical compromise that already provides very low short-term noise while improving operational efficiency and reducing the consumption of both reference and working-standard gases. At Jurong, where no drying was applied, reference gases became humidified in the sampling lines; these values were first corrected for H2O interference before calibration. A detailed description of all reference gases is given in Table 1.
For the assigned reference-gas values (Ref1–3), CH4 mole fractions were reported on the WMO X2004A scale and δ13CH4 values were reported relative to the VPDB scale. The remaining working standard gases (WS1–WS5) were assigned in our laboratory by calibration against Ref1–3, thereby tying our working scale to the INSTAAR laboratory scale. The working standard gases were calibrated using the isotopologue-specific approach. Section 2.3 then describes two alternative strategies for applying these assigned values to calibrate and correct the laboratory and field measurements. To assess calibration performance, mid-level references (WS2 at Jurong and Ref2 in the laboratory) were treated as “targets”, while the remaining references were used for calibration. Correction coefficients derived from linear interpolation between reference and measured values were then applied to the target periods.
All reference gas values used in this study, across both laboratory and field experiments, were traceable to internationally recognized calibration scales. CH4 mole fractions were reported on the World Meteorological Organization (WMO) X2004A scale. δ13CH4 values were reported relative to the Vienna Pee Dee Belemnite (VPDB) scale and were linked through assigned reference-gas values, including manufacturer-certified values for Ref1–Ref3 and laboratory-assigned values for the remaining reference gases using working standards linked to the Institute of Arctic and Alpine Research (INSTAAR, University of Colorado Boulder) laboratory scale. Section 2.3 then describes two alternative strategies for applying these assigned reference-gas values to calibrate and correct the laboratory and field measurements.
2.3 Calibration and correction strategies
Following the water vapor correction, two calibration strategies were applied to derive δ13CH4 from the analyzer outputs, which were the isotopologue-specific calibration approach (Wen et al., 2013; Griffith, 2018) and the direct δ13CH4 calibration method (delta-based). These methods were evaluated under both dry and humidified reference gas conditions.
2.3.1 Isotopologue-specific calibration
This method involves two steps: (1) Separate linear correction equations were established for 12CH4 and 13CH4 using two reference gases with significantly different isotopologue concentrations. Calibration coefficients a and b were determined from the linear relationship between calibrated and observed values. (2) These coefficients were subsequently applied to the target sample measurements to calculate the total CH4 concentration and δ13CH4 values using the following equations:
To perform the correction, the mole fractions of 12CH4 and 13CH4, as well as the δ13CH4, and δD-CH4 values of each reference gas, must be known. Here, 12CH4 and 13CH4 represent the measured mole fractions (ppb) of the two carbon isotopologues of CH4, obtained directly from the spectrometer under dry air conditions. CH4 in Eq. (4) denotes the total methane mole fraction, i.e., the sum of all isotopologues. δ13CH4 (‰, VPDB scale) and δD–CH4 (‰, Vienna Standard Mean Ocean Water, VSMOW scale) denote the carbon and hydrogen isotopic compositions of methane, with values reported in per mil (‰), respectively, determined based on certified reference gases or previous intercomparison results. Constants used in the calculations included 13rref=0.0111802 (VPDB) and 2rref=0.00015575 (VSMOW), which are the internationally accepted reference isotope ratios for 13C 12C and , respectively, as recommended by Werner and Brand (2001).
The parameters a12, b12, a13, and b13 represent the slope and intercept of the calibration equations for 12CH4 and 13CH4, respectively. They correct the instrument response and convert the raw isotopologue signals into calibrated mole fractions. For field processing, these coefficients were linearly interpolated in time between calibration periods, rather than applying an interval-mean approximation, to obtain time-resolved calibration parameters for each measurement. The isotope ratio 13r is calculated directly from the calibrated isotopologue mole fractions, and the total isotopologue normalization factor Rsum accounts for all possible isotopic substitutions in CH4, including hydrogen-bearing species such as CH3D, following Griffith (2018). An assumed δD value of −100 ‰ for atmospheric CH4 was adopted from Quay et al. (1999). All calibrated values were reported on the WMO X2004A scale, and all isotopic ratios are reported relative to the VPDB (for δ13CH4) and VSMOW (for δD) international reference scales.
A sensitivity estimate was conducted to assess the impact of uncertainty in the assumed atmospheric δD–CH4 on Rsum and total CH4 mole fraction. Taking δD–CH ‰ as a representative value, a conservative ±50 % variation (−50 ‰ to −150 ‰) changes 2r by ±5.6 %, but propagates to Rsum in Eq. (6) at only ±0.0031 %. This corresponds to <0.1 ppb uncertainty in total CH4 for typical atmospheric mole fractions of 2000–3000 ppb, which is negligible relative to measurement uncertainty. Note that Rsum is used only to convert 12CH4 to total CH4 and does not enter the calculation of δ13CH4, which is determined from the ratio 13CHCH4.
In this study, all humidity-correction functions were derived from the raw (uncorrected) outputs, and all subsequent analyses were performed using the raw 12CH4, 13CH4, and δ13CH4 values with the offline empirical humidity correction developed here. The manufacturer-corrected outputs were not used in the calibration workflow to avoid double correction.
2.3.2 Delta-based calibration
The delta-based calibration approach corrects instrumental drift using δ13CH4 directly. A linear calibration is established using two reference gases with distinct δ13CH4 signatures as Eq. (8):
Here, aδ and bδ represent the slope and intercept of the delta-based calibration, respectively, which account for small systematic differences between the spectrometer-derived δ13CH4 values and the true isotopic compositions of the reference gases. In both field measurements, the two reference gases used for the delta-based calibration had very similar δ13CH4 values. Therefore, aδ was effectively set to 1 and the delta-based calibration was implemented as a one-point correction. These coefficients correct residual scale offsets and sensitivity deviations in the δ13CH4 retrievals before converting all measurements to the VPDB scale. This calibration is then applied to field observations, and δ13CH4 values are reported on the VPDB scale.
2.4 Correlation and statistics analysis
All statistical analyses were performed with a significance threshold of p<0.05. Uncertainties were expressed as 95 % confidence intervals derived from bootstrap resampling. To assess the error characteristics, residuals and inter-method differences were visualized as histograms and fitted with Gaussian functions. The mean, standard deviation (σ), RMSE, and MAE were computed to characterize the residual statistics. Gaussian functions were fitted to the histograms to examine whether the residuals followed a normal distribution.
3.1 Water vapor correction functions
We first performed laboratory experiments to quantify the water vapor effect on isotopologue mole fractions and δ13CH4. Reference gases with known CH4 mole fractions and δ13CH4 values were humidified to obtain varying H2O levels. The experiments revealed systematic dependencies of the 12CH4 and 13CH4 mole fractions and δ13CH4, on H2O concentrations (Fig. 3a–c).
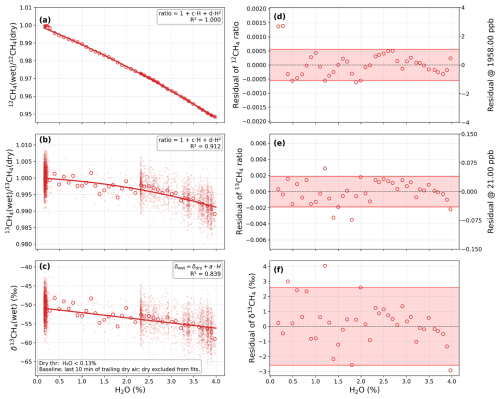
Figure 3Water vapor correction functions for 12CH4, 13CH4, and δ13CH4 derived from laboratory water vapor experiments (0.15 %–4.0 % H2O). Panels (a), (b), and (c) show the fitted relationships between H2O concentration and the wet-to-dry ratios of 12CH4, the wet-to-dry ratios of 13CH4, and the δ13CH4 deviation (δ13CH4, wet – δ13CH4, dry), respectively. Each open circle represents the mean value within a 0.1 % H2O bin. The solid red lines indicate the best-fit regression models (quadratic for 12CH4 and 13CH4, linear for δ13CH4), and small points indicate raw observations. Panels (d), (e), and (f) show the corresponding residuals; the red shaded bands indicate the interval containing 90 % of the data points, with parameters detailed in Table 3.
From these measurements, we established quadratic polynomial correction functions to describe the H2O dependence of isotopologue mole fractions (Eqs. 9–10), and derived an empirical linear function for δ13CH4 (Eq. 11). These relationships form the foundation for the water correction in field evaluations of calibration strategies (Sect. 3.3).
Here, the “wet” mole fractions represent values observed from the humidified reference gas in laboratory conditions, while the “dry” values correspond to the instrument-measured dry baseline. The baseline was obtained from the low-humidity segment at the end of the experiment (H2O < 0.13 %). H2O denotes the water vapor concentration (%) directly measured by the analyzer (column “H2O”). For Eq. (9), the quadratic fit for 12CH4 is robust (R2≈1.000), with fitted uncertainties of for the linear term and for the quadratic term. For Eq. (10), the quadratic dependence for 13CH4 is also significant (R2≈0.912), and the fitted uncertainties are and for the linear and quadratic terms, respectively. For Eq. (11), δ13CH4, wet exhibits a linear dependence on water vapor concentration, deviating from the dry reference δ13CH4, dry by ‰ per % H2O (R2≈0.839).
Table 3Summary statistics of water vapor correction residuals for 12CH4, 13CH4, and δ13CH4.

Residuals were calculated as the absolute difference between binned means and fitted values from the water vapor correction functions for the experiment (0.15 %–4.0 % H2O). Reported statistics include minimum (Min), median, 80th and 90th percentile (80th percentile and 90th percentile), maximum (Max), and Root Mean Square (RMS) residuals. Percentile metrics (P80, P90) are used to represent the typical residual range while minimizing the influence of a few extreme humid points. All values are absolute residuals; 12CH4 and 13CH4 are in ppb, and δ13CH4 in ‰.
The residual statistics of the fitted water vapor correction functions are shown in Fig. 3d–f, and detailed summarized in Table 3. The absolute residuals between the binned means and the fitted values were mostly small, confirming that the empirical corrections effectively capture the water vapor dependencies within 0.15 %–4.0 % H2O. For 12CH4, 80 % of the absolute residuals were below 0.93 ppb and 90 % below 1.09 ppb, corresponding to less than 0.05 % of the 12CH4 reference concentration. The 13CH4 residuals were similarly low, with 80 % and 90 % below 0.033 and 0.041 ppb (median = 0.021 ppb, RMS = 0.029 ppb). For δ13CH4, the median and 80th percentile residuals were 0.63 ‰ and 1.68 ‰, respectively. Both 12CH4 and 13CH4 required quadratic correction functions to accurately describe the nonlinear response to water vapor, reflecting the combined influence of dilution and pressure-broadening effects on observed absorption peak heights. This nonlinear behavior is consistent with previous characterizations of CRDS instruments (Chen et al., 2010; Wen et al., 2013; Griffith, 2018).
For 12CH4, the fitted coefficients (−0.0103 for the linear and for the quadratic term) closely match those reported by Chen et al. (2010) for bulk CH4, confirming the reproducibility of the water vapor interference across different analyzer models and laboratory setups. For 13CH4, the same quadratic correction was applied. Although the absolute residuals are small (80th percentile = 0.033 ppb), the fractional deviation is larger than for 12CH4 because the 13CH4 dry mole fraction is only ∼21 ppb. Quantitatively, the 80th percentile residual corresponds to a relative uncertainty of approximately 0.15 % for 13CH4, compared with ∼0.05 % for 12CH4, indicating that the 13CH4 channel has about three times higher relative uncertainty. This suggests that the 13CH4 signal is intrinsically more vulnerable to residual water bias. Because δ13CH4 is derived from the ratio of 13CH4 to 12CH4, any remaining humidity-dependent bias in 13CH4 directly propagates into δ13CH4. In practice, this means that the accuracy of the isotopologue-specific calibration strategy under humid conditions is ultimately limited by the performance of the 13CH4 water vapor correction.
By contrast, δ13CH4 exhibited an approximately linear dependence on water vapor. This behavior arises because the nonlinear contributions in the numerator and denominator largely cancel when expressed as a ratio, leaving a dominant first-order term. This partial cancellation of nonlinear terms reflects the mathematical structure of δ13CH4 as a ratio, where similar H2O dependencies in 12CH4 and 13CH4 tend to offset each other. Although the δ13CH4-H2O regression shows larger scatter than those for the individual isotopologues, the fitted slope of ‰ %−1, H2O over the full experimental range of 0.15–4.0 % H2O reflects enhanced sensitivity at higher humidity levels. When restricted to the same water vapor interval (≤1.5 %), our fitted slope of ‰ %−1 H2O is not significantly different from the reported slope of ‰ %−1 H2O by Hoheisel et al. (2019).
It is worth noting that physical drying to approximately 0.1 % H2O remains the recommended best practice for achieving sub-per-mil δ13CH4 accuracy (e.g., Rella et al., 2015). In our study, the empirical water vapor correction functions were derived for the H2O range of 0.15 %–4.0 %, which provide a complementary solution for measurements in humid air.
3.2 Comparison of δ13CH4 calibration strategies for dry air sample measurements
For dry air sample measurements, both the isotopologue-specific and the delta-based calibration strategies yielded consistent δ13CH4 results, with small and relatively stable offsets. For the certified target gas (Fig. 4), both calibrated values were close to the assigned reference, with mean residuals of 0.15 ‰ for the isotopologue-specific method and 0.55 ‰ for the delta-based method. The inter-method difference (Δδ13CH4= iso – delta) averaged −0.40 ‰, indicating a slight but systematic offset toward lighter δ13C in the delta-based calibration. Residuals and differences followed approximately normal distributions, and their root-mean-square and mean absolute errors were both close to 2 ‰, comparable to the typical instrumental precision of the analyzer.
For measurements of air samples dried with the Nafion™ membrane dryer at the SORPES station (Fig. 5), both calibration schemes showed highly consistent δ13CH4 retrievals. The two approaches yielded overlapping daily means throughout DOY 240–280, with differences mostly within the 1σ range of observational variability. The inter-method difference (Δδ13CH4) averaged 0.29 ‰ and exhibited a near-Gaussian distribution (σ=0.71 ‰), indicating minimal systematic bias between the two schemes for dried air sample measurements. A significant correlation between Δδ13CH4 and CH4 mole fraction (R2=0.97, p<0.001) suggests that part of the residual offset may result from concentration-dependent effects of delta-based calibration. Although the ambient data showed slightly higher variability than laboratory measurements, both calibration methods remained stable and consistent across a wide range of CH4 concentrations, demonstrating reliable performance for dried-air applications.
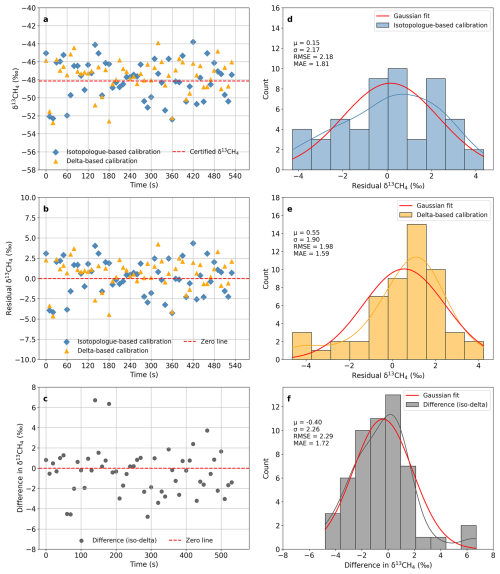
Figure 4Comparison of δ13CH4 calibration strategies for a dry reference gas. (a) Calibrated δ13CH4 from isotopologue-specific (blue diamonds) and delta-based (yellow triangles) strategies; the reference is shown as a red dashed line. (b) Residuals (calibrated – certified) for both strategies. (c) Time series of the difference between isotopologue-specific and delta-based calibrated δ13CH4 (hereafter referred to as the inter-method difference, iso – delta). (d, e) Histograms of residuals for the two strategies with Gaussian fits. (f) Distribution of the inter-method difference (iso – delta) with a Gaussian fit. For (d)–(f), Gaussian fits provide the mean (μ) and standard deviation (σ), while root-mean-square error (RMSE) and mean absolute error (MAE) are computed from the data. All results are based on 10-s averaged data.
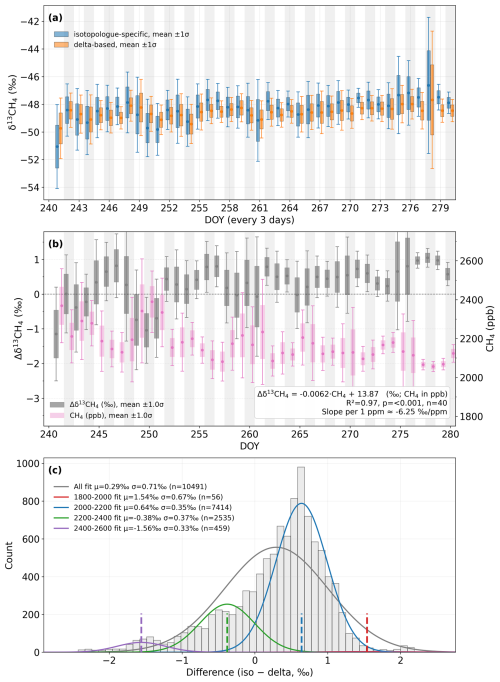
Figure 5Comparison of δ13CH4 calibration strategies at the SORPES station (dried ambient air, DOY 240–280, 2022). (a) Daily averaged δ13CH4 values from isotopologue-specific (blue) and delta-based (orange) calibration strategies, with 1σ standard deviations. (b) Time series of the inter-method difference Δδ13CH4 (iso – delta, grey bars) and corresponding CH4 mole fraction (pink bars, right axis). The linear relationship between Δδ13CH4 and CH4 is shown with a fitted regression. (c) Histogram of Δδ13CH4 (iso − delta) for all data and for different CH4 concentration ranges, each fitted with a Gaussian function. The fitted mean (μ) and standard deviation (σ) are reported for each subset. All analyses are based on 5-min averaged data.
For dry air sample measurements, both calibration strategies yielded nearly identical δ13CH4 results, indicating that the isotopic retrievals are consistent when water vapor interference is negligible. However, a strong correlation between the inter-method difference (Δδ13CH4) and CH4 mole fraction (Fig. 5b) suggests that concentration-dependent effects may still influence the delta-based calibration. The Δδ13CH4 exhibits a clear and consistent dependence on CH4 mole fraction across the three concentration subsets (Fig. A2a–c). Examination of the two retrievals separately further indicates that both calibration formulations vary with CH4 mole fraction, but with different magnitudes and response patterns (Fig. A2d–f). The isotopologue-specific calibrated δ13CH4 shows a clearer CH4 dependence in all subsets (Fig. A2g–i), whereas the delta-based retrieval also varies with CH4 but generally with weaker explanatory power (lower R2) and a different slope structure (Fig. A2g–i). Thus, the concentration dependence of Δδ13CH4 reflects the different concentration sensitivities of the two calibration strategies and their combined propagation into the inter-method difference.
Although the present dataset cannot explicitly isolate the cause, this pattern agrees well with the theoretical framework of Griffith et al. (2012), which demonstrated that non-zero intercepts and nonlinearities in isotopologue calibrations inevitably propagate into δ13C space, producing apparent δ13C-concentration coupling. Griffith (2018) further generalized this analysis, identifying both inverse and linear dependencies of δ13CH4 on CH4 concentration. Our results exhibit the concentration-dependent behavior predicted by these studies. Similar dependencies have also been reported for CO2 isotope measurements (Wen et al., 2013; Pang et al., 2016; Braden-Behrens et al., 2017), indicating that such effects are intrinsic to δ13C-based formulations rather than instrument-specific anomalies. Beyond the dry air regime examined here, additional deviations may emerge in humid air, where spectral interference becomes a dominant factor influencing isotopic accuracy.
The concentration dependence diagnosed here suggests that, in future deployments, CH4 concentration dependence could be incorporated directly into the delta-based calibration. With a set of at least 3 reference gases covering a reasonable CH4 concentration range, the linear delta-based calibration (Eq. 8) could be replaced with a 3-parameter equation presented in Eq. (15) of Griffith (2018), without requiring a spread in δ13CH4 values across the reference gases. To further assess the robustness of these correction functions under realistic environmental conditions, we examined their performance in humidified air samples at the rice paddy site.
3.3 Performance of calibration strategies under humid air conditions
To evaluate the performance of the water vapor correction under field conditions, both calibration strategies were applied to a humidified target gas at the rice paddy site (DOY 240–280, 2018). As shown in Fig. 6, after applying the correction functions, both isotopologue-specific and delta-based calibrations reproduced δ13CH4 values close to the certified reference (Fig. 6a), confirming that the equations effectively removed humidity-induced artifacts. The isotopologue-specific calibration yields δ13CH4 values that align more tightly with the reference, while the delta-based calibrated results retain a small positive offset (Fig. 6a–c). Consistent with the histograms, the isotopologue-specific residuals improved from ‰ (σ=0.30 ‰) before correction to ‰ (σ=0.24 ‰) after correction. In contrast, the delta-based residuals remained consistently near +0.5 ‰ both before and after correction ( to +0.49 ‰, σ=0.26 ‰; Fig. 6d–e). The inter-method difference (iso – delta) has a mean of −0.80 ‰ after correction with a reduced spread (σ=0.14 ‰; Fig. 6f), indicating a stable, strategy-dependent offset. These results confirm that the correction equations effectively mitigate humidity-induced artifacts, particularly for the isotopologue-specific approach, providing a reliable basis for subsequent field validation.
The Jurong site represents a typical rice paddy ecosystem, characterized by persistently high ambient humidity and strong methane emissions. During the observation period, H2O concentrations frequently exceeded 3 %, while CH4 mole fractions varied substantially, ranging from background levels below 2000 ppb to episodic peaks above 5000 ppb. These conditions provide a stringent test for calibration strategies, as both elevated humidity and broad concentration ranges can amplify systematic biases in δ13CH4 retrievals.
Figure 7 illustrates the contrasting behavior of isotopologue-specific and delta-based calibrations for humid air observations. Daily mean δ13CH4 values from the delta-based calibration were consistently offset relative to those from the isotopologue-specific approach, and exhibit greater variability (Fig. 7a). The inter-method difference, Δδ13CH4 (iso − delta), closely tracked temporal variations in both H2O and CH4 mole fractions (Fig. 7b), exhibiting significant correlations with H2O (R2=0.91, p<0.001) and with CH4 (R2=0.28, p<0.001). The overall Δδ13CH4 distribution is centered at −1.06 ‰ (σ=1.92 ‰; n=3126) (Fig. 7c). This bias varies with CH4 concentration, and the fitted mean Δδ13CH4 shifts from −1.55 ‰ (2000–2500 ppb) to +2.20 ‰ (5000–5500 ppb) (Fig. 7c).
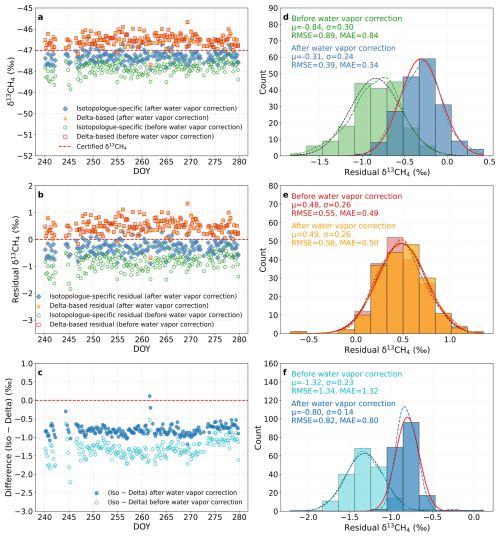
Figure 6Comparison of δ13CH4 calibration strategies for a humidified target gas (rice paddy site, DOY 240–280, 2018). (a) δ13CH4 values from the isotopologue-specific (blue diamonds) and delta-based (orange triangles) calibrations, shown both before (open symbols) and after (filled symbols) applying the water vapor correction. The certified reference value is indicated by the red dashed line. (b) Residuals (calibrated – certified) for both calibration strategies, with open and filled markers representing results before and after water vapor correction, respectively. (c) Time series of the inter-method difference Δδ13CH4= (iso – delta), shown before (open) and after (filled) water vapor correction. (d, e) Histograms of residuals for the two calibration strategies before and after water vapor correction, with Gaussian fits illustrating their respective distributions. (f) Histogram of the inter-method difference (iso − delta) before and after water vapor correction, also fitted with Gaussian functions. For panels (d)–(f), Gaussian fits provide the mean (μ) and standard deviation (σ), while the root-mean-square error (RMSE) and mean absolute error (MAE) are calculated from the data. All results are based on 5-min averaged measurements after applying the empirical water vapor correction equations functions.

Figure 7Field comparison of δ13CH4 calibration strategies for humid air observations at the rice paddy site (DOY 240–280, 2018). (a) Daily mean δ13CH4 from isotopologue-specific (blue) and delta-based (orange) calibrations (mean ±1σ). (b) Time series of the inter-method difference in δ13CH4 (iso – delta, blue line) together with total CH4 mole fraction (orange line) and H2O concentration (green line, 0.93 % to 3.5 %). (c) Histogram of δ13CH4 differences (iso – delta) grouped by CH4 mole-fraction ranges, each fitted with a Gaussian function. The fitted mean (μ) and standard deviation (σ) are reported for each subset. All results are based on 5-min averages.
To further interpret Fig. 7, we added the Jurong correlation analysis in Fig. A1. The inter-method difference (Δδ13CH4) shows only a weak relationship with CH4 mole fraction (R2≈0.30) and is not statistically significant (p>0.05; Fig. A1a), which may reflect the intrinsic concentration dependence for the delta-based approach has been reported previously (Rella et al., 2015; Miles et al., 2018).
In contrast, Δδ13CH4 correlates strongly with H2O (R2≈0.91, p<0.001; Fig. A1b). This is most likely driven by the significant correlation between the calibrated δ13CH4 using the delta-based approach and H2O (Fig. A1d), as the correlation between the calibrated δ13CH4 using the isotopologue-specific approach and H2O is insignificant (Fig. A1c).
These results highlight that high humidity and large CH4 variability exacerbate the intrinsic weaknesses of delta-based calibration, consistent with earlier observations for CO2 isotopes (Wen et al., 2013; Pang et al., 2016). Without explicit correction, such biases can propagate into source signature retrievals, leading to systematic offsets in rice-paddy plume analyses. Although a significant correlation between Δδ13CH4 and H2O was observed, this may not directly represent a spectroscopic effect of water vapor. At the Jurong site, episodes of high humidity often coincided with strong CH4 emissions, producing an intrinsic covariance between H2O and CH4. Consequently, part of the apparent Δδ13CH4–H2O relationship may arise from this co-variation rather than from water vapor interference alone. The empirical correction functions used here effectively capture the dominant humidity effects at the isotopologue level, but may not fully account for additional spectral interferences associated with pressure or temperature variability. After accounting for the H2O–CH4 covariance, the remaining concentration dependence is most consistent with intrinsic CH4 non-linearity reported for CRDS isotope measurements (e.g., Rella et al., 2015; Miles et al., 2018), while residual pressure/temperature-related spectral effects and limitations of empirical corrections may also contribute. We therefore interpret the field results as a combination of effectively corrected first-order humidity effects and residual concentration-dependent behavior that is instrument- and condition-dependent. It should also be emphasized that, in practice, physical drying of air samples remains the preferred approach, whereas empirical water vapor corrections should serve only as a secondary option when drying is not feasible. Addressing these aspects in future work will further refine calibration strategies and enhance their applicability across diverse environments.
3.4 Application of calibration strategies in field observations
Jurong is situated in rice paddy fields and is therefore best characterized as a large-signal, source-influenced environment, and we refer to the WMO/GAW extended compatibility target of ±0.2 ‰ for the target accuracy of our measurements (IAEA, 2024). In contrast, SORPES captures both well-mixed background periods and large-signal episodes, so the appropriate accuracy requirement depends on the specific application and is expected to fall between the WMO/GAW network target (±0.02 ‰, for well-mixed background air) and the extended target (±0.2 ‰, for large-signal (source-influenced/urban) conditions). Our target-gas tests suggest that the isotopologue-specific approach can satisfy the ±0.2 ‰ criterion under dry conditions, and that applying an effective water-vapor correction brings performance close to this extended target under humid conditions. However, in both cases, they do not meet the stringent ±0.02 ‰ compatibility level required for background-network applications. Achieving that level will likely require improved instrumental precision and stability, together with sufficiently frequent calibration to reference gases.
To further quantify the relationship between the inter-method bias (Δδ13CH4) and CH4 concentration, we regressed Δδ13CH4 against CH4 mole fraction using the expression Δδ13CH + c2× CH4 following Griffith et al. (2018) (Table 4). At both field sites, most data points are concentrated within a relatively narrow CH4 range, representing the dominant concentration regimes during field observations: 2000–3000 ppb at Jurong and 2000–2500 ppb at SORPES. These ranges capture the typical operational conditions under which calibration biases are most relevant.
At the Jurong site, Δδ13CH4 exhibited a pronounced dependence, with significantly negative c1 terms (p<0.01) and negligible c2 values. This pattern indicates that the inter-method difference increases toward lower CH4 concentrations, where signal dilution and pressure-broadening effects become more influential. Although the water vapor correction successfully mitigates first-order humidity effects, the remaining dependence likely arises from the coupled response of isotopologue scaling and dilution to varying CH4 and H2O levels. At the SORPES site, Δδ13CH4 was better characterized by the CH4 (c2) term, showing a weak but positive dependence on concentration (c2>0, p<0.05). In the absence of strong humidity effects, the difference between isotopologue-specific and delta-based calibrations thus reflects higher-order nonlinearities intrinsic to the delta-based formulation. The contrasting dominant terms between Jurong and SORPES highlight how humidity modulates the expression of calibration nonlinearity: humid air amplifies inverse () dependencies, whereas dry air emphasizes minor linear (CH4) effects.
These site-specific regressions are consistent with the theoretical framework proposed by Griffith (2018), where the inverse () and linear (CH4) terms correspond to intercept-driven and quadratic nonlinearities, respectively. The slope variations observed in Fig. 7c further support this interpretation, providing a mechanistic explanation for the site-dependent discrepancies in Figs. 6–7. However, despite the effectiveness of the water vapor correction functions across the full humidity range, the residual concentration dependence of Δδ13CH4 suggests that both CH4 and H2O jointly modulate the inter-method bias, with their relative contributions differing between humid and dry air. In practice, field measurements typically include an air-drying stage (e.g., Nafion™ membrane dryers), but physical drying alone cannot fully remove water vapor interference. Even well-maintained Nafion™ systems leave residual H2O at 0.3 %–0.6 % under ambient conditions – enough to bias δ13CH4 retrievals, particularly at humid sites or during high-CH4 events. Therefore, an explicit H2O correction remains necessary rather than assuming that drying alone ensures isotopic accuracy (Welp et al., 2013; Paul et al., 2020).
Overall, these site-specific behaviors provide practical guidance for field deployment. Under well-dried conditions with relatively stable CH4 mole fractions (e.g., SORPES), the residual difference between the isotopologue-specific and delta-based calibrations is small, dominated by weak high-concentration nonlinearities, and can be characterized empirically. In such cases, the delta-based approach remains operationally acceptable for routine monitoring. By contrast, for humid air, lower CH4 conditions with large CH4 variability (e.g., Jurong), the inter-method bias exhibits a strong inverse-concentration dependence and should not be treated as a constant offset. In these environments, the isotopologue-specific calibration is required to avoid systematic shifts in inferred source signatures. In practice, physical drying of the sample air should remain the primary strategy wherever feasible, and the combination of humidity correction and isotopologue-specific calibration should be considered the default fallback when effective drying cannot be maintained.
Table 4Regression results of concentration dependence of Δδ13CH4 at Jurong (humid air) and SORPES (dry air) sites. Δδ13CH4 is defined as the inter-method difference between isotopologue-specific and delta-based calibrated δ13CH4 (Δ=δ13CH4_iso – δ13CH4_delta). The dependence on CH4 mole fraction was fitted using the function CH4 + c2CH4, where c0, c1, and c2 are regression coefficients (Griffth et al., 2018). All results are based on 5-min averaged data. Significance levels: p<0.05 (*), p<0.01 (**). The “Dominant term” column identifies whether the contribution of c1 or c2 is greater, based on their evaluated magnitudes at the median CH4 mole fraction of each concentration range. Bold rows indicate the concentration subset containing the highest proportion of observations for each site, and thus the most representative range for that site.

We evaluated two calibration strategies for δ13CH4, delta-based and isotopologue-specific calibration, using a Picarro G2201-i isotopic analyzer under both laboratory and field conditions. Empirical water vapor correction functions were established based on laboratory experiments (0.15 %–4.0 % H2O) to effectively remove humidity-induced biases in isotopologue mole fractions and δ13CH4. The observed water vapor dependencies were best represented by quadratic functions for 12CH4 and 13CH4, and a linear function for δ13CH4. For 12CH4 and 13CH4, the residuals fell within the analyzer's precision, while δ13CH4 residuals remained small and comparable to the precision. These water correction equations provide a robust basis for correcting field data. While most field systems employ physical air drying (e.g., Nafion™ dryers), residual H2O often persists at levels sufficient to introduce measurable isotopic bias. Therefore, explicit humidity correction remains necessary, particularly under high-humidity conditions.
For dry air measurements, both calibration strategies yielded consistent δ13CH4 results. Laboratory tests and dried-air observations at the SORPES site confirmed nearly identical retrievals between the two approaches, with a bias of 0.29 ‰, which is small but non-negligible relative to the extended compatibility goal (±0.2 ‰, k=2) and comparable to our target-gas repeatability. In contrast, significant inter-method discrepancies emerged for humid air measurement. The bias (Δδ13CH4) correlated strongly with both CH4 and H2O levels, indicating that humidity and concentration jointly modulate calibration accuracy. Consequently, the isotopologue-specific calibration method is better suited for accurate δ13CH4 retrievals under conditions of fluctuating humidity and CH4 concentrations.
The empirical water-vapor correction functions and calibration parameters reported here were derived for a single Picarro G2201-i analyzer and therefore should not be assumed to be directly transferable across instruments or deployments. Instrument-to-instrument differences (e.g., optical alignment, detector response, and aging) may lead to different humidity sensitivities and concentration dependences, so the empirical coefficients generally need to be re-derived and validated for each analyzer and field campaign over the relevant H2O and CH4 ranges. While the function and workflow are transferable, applying this framework to other instruments requires instrument-specific characterization using controlled humidity tests and routine reference-/target-gas checks.

Figure A1Correlations between δ13CH4 calibration results and CH4 and H2O for humid air observations at the Jurong site. Relationship between (a) Δδ13CH4 and CH4; (b) Δδ13CH4 and H2O; (c) isotopologue-specific δ13CH4 and H2O; (d) delta-based δ13CH4 and H2O. Dashed lines indicate fitted regressions. The fitted equations, R2, p, and n are reported in each panel.
The data used in this study are available from the corresponding author upon reasonable request.
J.L. and H.C. conceived the study. J.L. performed the laboratory and field experiments, analyzed the data. X.C. and A.D., W.J., Y.Z., and J.C. provided scientific guidance for the field experiments. J.L. and H.C. wrote the manuscript with contributions from all authors.
At least one of the (co-)authors is a member of the editorial board of Atmospheric Measurement Techniques.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
This article is part of the special issue “Greenhouse gas monitoring in the Asia–Pacific region (ACP/AMT/GMD inter-journal SI)”. It is a result of the 4th China Greenhouse Gas Monitoring Symposium, Nanjing, China, 2–3 November 2024.
The study was supported by the National Natural Science Foundation of China (grant numbers 42305129, 42475115, U24A20590), the Fundamental Research Funds for the Central Universities – Cemac “GeoX” Interdisciplinary Program (grant numbers 2024QNXZ01, 2024300245, 14380235). This work was supported by the project of Youth Crossdisciplinary Team of the Chinese Academy of Sciences (No. 2023000126). This research was also supported by the Collaborative Innovation Center of Climate Change, Jiangsu Province.
The study was supported by the National Natural Science Foundation of China (grant nos. 42305129, 42475115, U24A20590), the Fundamental Research Funds for the Central Universities – Cemac “GeoX” Interdisciplinary Program (grant nos. 2024QNXZ01, 2024300245, 14380235). This work was supported by the project of Youth Crossdisciplinary Team of the Chinese Academy of Sciences (grant no. 2023000126). This research was also supported by the Collaborative Innovation Center of Climate Change, Jiangsu Province.
This paper was edited by Andre Butz and reviewed by David Griffith and one anonymous referee.
Bakkaloglu, S., Lowry, D., Fisher, R. E., Menoud, M., Lanoisellé, M., Chen, H., Röckmann, T., and Nisbet, E. G.: Stable isotopic signatures of methane from waste sources through atmospheric measurements, Atmos. Environ., 276, 119021, https://doi.org/10.1016/j.atmosenv.2022.119021, 2022.
Berden, G. and Engeln, R.: Cavity ring-down spectroscopy: techniques and applications, John Wiley & Sons, https://doi.org/10.1002/9781444308259, 2009.
Braden-Behrens, J., Yan, Y., and Knohl, A.: A new instrument for stable isotope measurements of 13C and 18O in CO2 – instrument performance and ecological application of the Delta Ray IRIS analyzer, Atmos. Meas. Tech., 10, 4537–4560, https://doi.org/10.5194/amt-10-4537-2017, 2017.
Chen, H., Winderlich, J., Gerbig, C., Hoefer, A., Rella, C. W., Crosson, E. R., Van Pelt, A. D., Steinbach, J., Kolle, O., Beck, V., Daube, B. C., Gottlieb, E. W., Chow, V. Y., Santoni, G. W., and Wofsy, S. C.: High-accuracy continuous airborne measurements of greenhouse gases (CO2 and CH4) using the cavity ring-down spectroscopy (CRDS) technique, Atmos. Meas. Tech., 3, 375–386, https://doi.org/10.5194/amt-3-375-2010, 2010.
Crosson, E.: A cavity ring-down analyzer for measuring atmospheric levels of methane, carbon dioxide, and water vapor, Appl. Phys. B, 92, 403–408, https://doi.org/10.1007/s00340-008-3135-y, 2008
Crosson, E. R., Ricci, K. N., Richman, B. A., Chilese, F. C., Owano, T. G., Provencal, R. A., Todd, M. W., Glasser, J., Kachanov, A. A., and Paldus, B. A.: Stable isotope ratios using cavity ring-down spectroscopy: determination of 13C/12C for carbon dioxide in human breath, Anal. Chem., 74, 2003–2007, https://doi.org/10.1021/ac025511d, 2002.
Defratyka, S. M., France, J. L., Fisher, R. E., Lowry, D., Fernandez, J. M., Bakkaloglu, S., Yver-Kwok, C., Paris, J.-D., Bousquet, P., and Arnold, T.: Evaluation of data processing strategies for methane isotopic signatures determined during near-source measurements, Tellus B, 77, https://doi.org/10.16993/tellusb.1878, 2025.
De Groot, P. A.: Handbook of stable isotope analytical techniques, Elsevier, https://www.sciencedirect.com/science/book/9780444511140 (last access: 9 March 2026), 2004.
Ding, A., Nie, W., Huang, X., Chi, X., Sun, J., Kerminen, V.-M., Xu, Z., Guo, W., Petäjä, T., Yang, X., Kulmala, M., and Fu, C.: Long-term observation of air pollution–weather/climate interactions at the SORPES station: a review and outlook, Frontiers of Environmental Science & Engineering, 10, 15, https://doi.org/10.1007/s11783-016-0877-3, 2016.
Ehleringer, J. R. and Osmond, C. B.: Stable isotopes, in: Plant physiological ecology: field methods and instrumentation, Springer, 281–300, https://doi.org/10.1007/978-94-009-2221-1, 1989.
Flores, E., Viallon, J., Moussay, P., Griffith, D. W., and Wielgosz, R. I.: Calibration strategies for FT-IR and other isotope ratio infrared spectrometer instruments for accurate δ13C and δ18O measurements of CO2 in air, Anal. Chem., 89, 3648–3655, https://doi.org/10.1021/acs.analchem.6b05063, 2017.
Forster, P., Storelvmo, T., Armour, K., Collins, W., Dufresne, J.-L., Frame, D., Lunt, D., Mauritsen, T., Palmer, M., and Watanabe, M.: The Earth's energy budget, climate feedbacks, and climate sensitivity, in: Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, pp. 923–1054, https://doi.org/10.1017/9781009157896.009, 2021.
France, J. L., Fisher, R. E., Lowry, D., Allen, G., Andrade, M. F., Bauguitte, S. J.-B., Bower, K., Broderick, T. J., Daly, M. C., and Forster, G.: δ13C methane source signatures from tropical wetland and rice field emissions, Philos. T. R. Soc. A, 380, 20200449, https://doi.org/10.1098/rsta.2020.0449, 2022.
Griffith, D. W. T.: Calibration of isotopologue-specific optical trace gas analysers: a practical guide, Atmos. Meas. Tech., 11, 6189–6201, https://doi.org/10.5194/amt-11-6189-2018, 2018.
Griffith, D. W. T., Deutscher, N. M., Caldow, C., Kettlewell, G., Riggenbach, M., and Hammer, S.: A Fourier transform infrared trace gas and isotope analyser for atmospheric applications, Atmos. Meas. Tech., 5, 2481–2498, https://doi.org/10.5194/amt-5-2481-2012, 2012.
Hoheisel, A., Yeman, C., Dinger, F., Eckhardt, H., and Schmidt, M.: An improved method for mobile characterisation of δ13CH4 source signatures and its application in Germany, Atmos. Meas. Tech., 12, 1123–1139, https://doi.org/10.5194/amt-12-1123-2019, 2019.
IAEA: Measurement of the stable carbon isotope ratio in atmospheric CH4 using laser spectroscopy for CH4 source characterization, IAEA Pan. P., https://doi.org/10.61092/iaea.logm-wiux, 2024.
IPCC: Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, 996 pp., https://wedocs.unep.org/20.500.11822/30763 (last access: 9 March 2026), 2007.
Kirschke, S., Bousquet, P., Ciais, P., Saunois, M., Canadell, J. G., Dlugokencky, E. J., Bergamaschi, P., Bergmann, D., Blake, D. R., and Bruhwiler, L.: Three decades of global methane sources and sinks, Nat. Geosci., 6, 813–823, https://doi.org/10.1038/ngeo1955, 2013.
Lan, X., Basu, S., Schwietzke, S., Bruhwiler, L. M., Dlugokencky, E. J., Michel, S. E., Sherwood, O. A., Tans, P. P., Thoning, K., and Etiope, G.: Improved constraints on global methane emissions and sinks using δ13CH4, Global Biogeochem. Cy., 35, e2021GB007000, https://doi.org/10.1029/2021GB007000, 2021.
Levin, I., Bergamaschi, P., Dörr, H., and Trapp, D.: Stable isotopic signature of methane from major sources in Germany, Chemosphere, 26, 161–177, https://doi.org/10.1016/0045-6535(93)90419-6, 1993.
Li, J., Zhang, Y., Gu, L., Li, Z., Li, J., Zhang, Q., Zhang, Z., and Song, L.: Seasonal variations in the relationship between sun-induced chlorophyll fluorescence and photosynthetic capacity from the leaf to canopy level in a rice crop, J. Exp. Bot., 71, 7179–7197, https://doi.org/10.1093/jxb/eraa408, 2020.
Miles, N. L., Martins, D. K., Richardson, S. J., Rella, C. W., Arata, C., Lauvaux, T., Davis, K. J., Barkley, Z. R., McKain, K., and Sweeney, C.: Calibration and field testing of cavity ring-down laser spectrometers measuring CH4, CO2, and δ13CH4 deployed on towers in the Marcellus Shale region, Atmos. Meas. Tech., 11, 1273–1295, https://doi.org/10.5194/amt-11-1273-2018, 2018.
Miller, J. B., Mack, K. A., Dissly, R., White, J. W., Dlugokencky, E. J., and Tans, P. P.: Development of analytical methods and measurements of 13C/12C in atmospheric CH4 from the NOAA Climate Monitoring and Diagnostics Laboratory global air sampling network, J. Geophys. Res.-Atmos., 107, ACH11-11–ACH11-15, https://doi.org/10.1029/2001JD000630, 2002.
Nisbet, E., Dlugokencky, E., Manning, M., Lowry, D., Fisher, R., France, J., Michel, S., Miller, J., White, J., and Vaughn, B.: Rising atmospheric methane: 2007–2014 growth and isotopic shift, Global Biogeochem. Cy., 30, 1356–1370, https://doi.org/10.1002/2016gb005406, 2016.
Nisbet, E., Fisher, R., Lowry, D., France, J., Allen, G., Bakkaloglu, S., Broderick, T., Cain, M., Coleman, M., and Fernandez, J.: Methane mitigation: methods to reduce emissions, on the path to the Paris agreement, Rev. Geophys., 58, e2019RG000675, https://doi.org/10.1029/2019RG000675, 2020.
Olivier, J. G. and Berdowski, J. J.: Global emission sources and sinks, in: The climate system, CRC Press, 33–77, https://doi.org/10.1201/9781003211266, 2021.
Pang, J., Wen, X., Sun, X., and Huang, K.: Intercomparison of two cavity ring-down spectroscopy analyzers for atmospheric 13CO2/12CO2 measurement, Atmos. Meas. Tech., 9, 3879–3891, https://doi.org/10.5194/amt-9-3879-2016, 2016.
Patra, P. K. and Khatri, P.: Atmospheric mixing ratio of greenhouse gases and radiative forcing, in: Handbook of Air Quality and Climate Change, Springer, 1–29, https://doi.org/10.1007/978-981-15-2527-8_29-1, 2022.
Paul, D., Scheeren, H. A., Jansen, H. G., Kers, B. A. M., Miller, J. B., Crotwell, A. M., Michel, S. E., Gatti, L. V., Domingues, L. G., Correia, C. S. C., Neves, R. A. L., Meijer, H. A. J., and Peters, W.: Evaluation of a field-deployable Nafion™-based air-drying system for collecting whole air samples and its application to stable isotope measurements of CO2, Atmos. Meas. Tech., 13, 4051–4064, https://doi.org/10.5194/amt-13-4051-2020, 2020.
Quay, P., Stutsman, J., Wilbur, D., Snover, A., Dlugokencky, E., and Brown, T.: The isotopic composition of atmospheric methane, Global Biogeochem. Cy., 13, 445–461, https://doi.org/10.1029/1998GB900006, 1999.
Rella, C.: Accurate stable carbon isotope ratio measurements in humid gas streams using the Picarro δ13CO2 G2101-i gas analyzer, White Paper of Picarro Inc., 1–13, https://www.picarro.com/sites/default/files/product_documents/White_Paper_G2101-i_Water_Correction.pdf (last access: 9 March 2026), 2012.
Rella, C. W., Chen, H., Andrews, A. E., Filges, A., Gerbig, C., Hatakka, J., Karion, A., Miles, N. L., Richardson, S. J., Steinbacher, M., Sweeney, C., Wastine, B., and Zellweger, C.: High accuracy measurements of dry mole fractions of carbon dioxide and methane in humid air, Atmos. Meas. Tech., 6, 837–860, https://doi.org/10.5194/amt-6-837-2013, 2013.
Rella, C. W., Hoffnagle, J., He, Y., and Tajima, S.: Local- and regional-scale measurements of CH4, δ13CH4, and C2H6 in the Uintah Basin using a mobile stable isotope analyzer, Atmos. Meas. Tech., 8, 4539–4559, https://doi.org/10.5194/amt-8-4539-2015, 2015.
Rice, A. L., Butenhoff, C. L., Teama, D. G., Röger, F. H., Khalil, M. A. K., and Rasmussen, R. A.: Atmospheric methane isotopic record favors fossil sources flat in 1980s and 1990s with recent increase, P. Natl. Acad. Sci. USA, 113, 10791–10796, https://doi.org/10.1073/pnas.1522923113, 2016.
Röckmann, T., Eyer, S., van der Veen, C., Popa, M. E., Tuzson, B., Monteil, G., Houweling, S., Harris, E., Brunner, D., Fischer, H., Zazzeri, G., Lowry, D., Nisbet, E. G., Brand, W. A., Necki, J. M., Emmenegger, L., and Mohn, J.: In situ observations of the isotopic composition of methane at the Cabauw tall tower site, Atmos. Chem. Phys., 16, 10469–10487, https://doi.org/10.5194/acp-16-10469-2016, 2016.
Saboya, E., Zazzeri, G., Graven, H., Manning, A. J., and Englund Michel, S.: Continuous CH4 and δ13CH4 measurements in London demonstrate under-reported natural gas leakage, Atmos. Chem. Phys., 22, 3595–3613, https://doi.org/10.5194/acp-22-3595-2022, 2022.
Saunois, M., Stavert, A. R., Poulter, B., Bousquet, P., Canadell, J. G., Jackson, R. B., Raymond, P. A., Dlugokencky, E. J., Houweling, S., Patra, P. K., Ciais, P., Arora, V. K., Bastviken, D., Bergamaschi, P., Blake, D. R., Brailsford, G., Bruhwiler, L., Carlson, K. M., Carrol, M., Castaldi, S., Chandra, N., Crevoisier, C., Crill, P. M., Covey, K., Curry, C. L., Etiope, G., Frankenberg, C., Gedney, N., Hegglin, M. I., Höglund-Isaksson, L., Hugelius, G., Ishizawa, M., Ito, A., Janssens-Maenhout, G., Jensen, K. M., Joos, F., Kleinen, T., Krummel, P. B., Langenfelds, R. L., Laruelle, G. G., Liu, L., Machida, T., Maksyutov, S., McDonald, K. C., McNorton, J., Miller, P. A., Melton, J. R., Morino, I., Müller, J., Murguia-Flores, F., Naik, V., Niwa, Y., Noce, S., O'Doherty, S., Parker, R. J., Peng, C., Peng, S., Peters, G. P., Prigent, C., Prinn, R., Ramonet, M., Regnier, P., Riley, W. J., Rosentreter, J. A., Segers, A., Simpson, I. J., Shi, H., Smith, S. J., Steele, L. P., Thornton, B. F., Tian, H., Tohjima, Y., Tubiello, F. N., Tsuruta, A., Viovy, N., Voulgarakis, A., Weber, T. S., van Weele, M., van der Werf, G. R., Weiss, R. F., Worthy, D., Wunch, D., Yin, Y., Yoshida, Y., Zhang, W., Zhang, Z., Zhao, Y., Zheng, B., Zhu, Q., Zhu, Q., and Zhuang, Q.: The Global Methane Budget 2000–2017, Earth Syst. Sci. Data, 12, 1561–1623, https://doi.org/10.5194/essd-12-1561-2020, 2020.
Saunois, M., Martinez, A., Poulter, B., Zhang, Z., Raymond, P. A., Regnier, P., Canadell, J. G., Jackson, R. B., Patra, P. K., Bousquet, P., Ciais, P., Dlugokencky, E. J., Lan, X., Allen, G. H., Bastviken, D., Beerling, D. J., Belikov, D. A., Blake, D. R., Castaldi, S., Crippa, M., Deemer, B. R., Dennison, F., Etiope, G., Gedney, N., Höglund-Isaksson, L., Holgerson, M. A., Hopcroft, P. O., Hugelius, G., Ito, A., Jain, A. K., Janardanan, R., Johnson, M. S., Kleinen, T., Krummel, P. B., Lauerwald, R., Li, T., Liu, X., McDonald, K. C., Melton, J. R., Mühle, J., Müller, J., Murguia-Flores, F., Niwa, Y., Noce, S., Pan, S., Parker, R. J., Peng, C., Ramonet, M., Riley, W. J., Rocher-Ros, G., Rosentreter, J. A., Sasakawa, M., Segers, A., Smith, S. J., Stanley, E. H., Thanwerdas, J., Tian, H., Tsuruta, A., Tubiello, F. N., Weber, T. S., van der Werf, G. R., Worthy, D. E. J., Xi, Y., Yoshida, Y., Zhang, W., Zheng, B., Zhu, Q., Zhu, Q., and Zhuang, Q.: Global Methane Budget 2000–2020, Earth Syst. Sci. Data, 17, 1873–1958, https://doi.org/10.5194/essd-17-1873-2025, 2025.
Schaefer, H., Whiticar, M. J., Brook, E. J., Petrenko, V. V., Ferretti, D. F., and Severinghaus, J. P.: Ice record of δ13C for atmospheric CH4 across the Younger Dryas–Preboreal transition, Science, 313, 1109–1112, https://doi.org/10.1126/science.1126562, 2006.
Schaefer, H., Fletcher, S. E. M., Veidt, C., Lassey, K. R., Brailsford, G. W., Bromley, T. M., Dlugokencky, E. J., Michel, S. E., Miller, J. B., and Levin, I.: A 21st-century shift from fossil-fuel to biogenic methane emissions indicated by 13CH4, Science, 352, 80–84, https://doi.org/10.1126/science.aad2705, 2016.
Shindell, D., Kuylenstierna, J. C., Vignati, E., van Dingenen, R., Amann, M., Klimont, Z., Anenberg, S. C., Muller, N., Janssens-Maenhout, G., and Raes, F.: Simultaneously mitigating near-term climate change and improving human health and food security, Science, 335, 183–189, https://doi.org/10.1126/science.1210026, 2012.
Tans, P. P., Crotwell, A. M., and Thoning, K. W.: Abundances of isotopologues and calibration of CO2 greenhouse gas measurements, Atmos. Meas. Tech., 10, 2669–2685, https://doi.org/10.5194/amt-10-2669-2017, 2017.
Tuzson, B., Mohn, J., Zeeman, M. J., Werner, R. A., Eugster, W., Zahniser, M. S., Nelson, D. D., McManus, J. B., and Emmenegger, L.: High precision and continuous field measurements of δ13C and δ18O in carbon dioxide with a cryogen-free QCLAS, Appl. Phys. B, 92, 451–458, https://doi.org/10.1007/s00340-008-3085-4, 2008.
Van Dingenen, R., Crippa, M., Maenhout, G., Guizzardi, D., and Dentener, F.: Global trends of methane emissions and their impacts on ozone concentrations, JRC Science for Policy Report, JRC113210, ISBN 978-92-79-96550-0, https://doi.org/10.2760/820175, 2018.
Wahl, E. H., Fidric, B., Rella, C. W., Koulikov, S., Kharlamov, B., Tan, S., Kachanov, A. A., Richman, B. A., Crosson, E. R., and Paldus, B. A.: Applications of cavity ring-down spectroscopy to high precision isotope ratio measurement of 13C/12C in carbon dioxide, Isot. Environ. Healt. S., 42, 21–35, https://doi.org/10.1080/10256010500502934, 2006.
Welp, L. R., Keeling, R. F., Weiss, R. F., Paplawsky, W., and Heckman, S.: Design and performance of a Nafion dryer for continuous operation at CO2 and CH4 air monitoring sites, Atmos. Meas. Tech., 6, 1217–1226, https://doi.org/10.5194/amt-6-1217-2013, 2013.
Wen, X.-F., Meng, Y., Zhang, X.-Y., Sun, X.-M., and Lee, X.: Evaluating calibration strategies for isotope ratio infrared spectroscopy for atmospheric 13CO2/12CO2 measurement, Atmos. Meas. Tech., 6, 1491–1501, https://doi.org/10.5194/amt-6-1491-2013, 2013.
Werner, R. A. and Brand, W. A.: Referencing strategies and techniques in stable isotope ratio analysis, Rapid Commun. Mass Sp., 15, 501–519, https://doi.org/10.1002/rcm.258, 2001.